Electrostatic and Structural Bases of Fe2+ Translocation through Ferritin Channels
- PMID: 27756844
- PMCID: PMC5207259
- DOI: 10.1074/jbc.M116.748046
Electrostatic and Structural Bases of Fe2+ Translocation through Ferritin Channels
Abstract
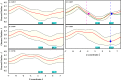
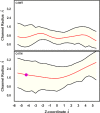
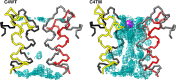
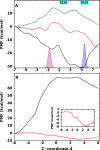
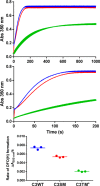
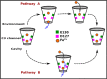
Ferritin molecular cages are marvelous 24-mer supramolecular architectures that enable massive iron storage (>2000 iron atoms) within their inner cavity. This cavity is connected to the outer environment by two channels at C3 and C4 symmetry axes of the assembly. Ferritins can also be exploited as carriers for in vivo imaging and therapeutic applications, owing to their capability to effectively protect synthetic non-endogenous agents within the cage cavity and deliver them to targeted tissue cells without stimulating adverse immune responses. Recently, X-ray crystal structures of Fe2+-loaded ferritins provided important information on the pathways followed by iron ions toward the ferritin cavity and the catalytic centers within the protein. However, the specific mechanisms enabling Fe2+ uptake through wild-type and mutant ferritin channels is largely unknown. To shed light on this question, we report extensive molecular dynamics simulations, site-directed mutagenesis, and kinetic measurements that characterize the transport properties and translocation mechanism of Fe2+ through the two ferritin channels, using the wild-type bullfrog Rana catesbeiana H' protein and some of its variants as case studies. We describe the structural features that determine Fe2+ translocation with atomistic detail, and we propose a putative mechanism for Fe2+ transport through the channel at the C3 symmetry axis, which is the only iron-permeable channel in vertebrate ferritins. Our findings have important implications for understanding how ion permeation occurs, and further how it may be controlled via purposely engineered channels for novel biomedical applications based on ferritin.
Keywords: ferritin; ferritin channels; ion channel; iron; iron uptake; molecular dynamics; multi-ion mechanism; structure; translocation.
© 2016 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures




References
-
- Arosio P., Carmona F., Gozzelino R., Maccarinelli F., and Poli M. (2015) The importance of eukaryotic ferritins in iron handling and cytoprotection. Biochem. J. 472, 1–15 - PubMed
-
- Theil E. C., Tosha T., and Behera R. K. (2016) Solving biology's iron chemistry problem with ferritin protein nanocages. Acc. Chem. Res. 49, 784–791 - PubMed
-
- Bou-Abdallah F. (2010) The iron redox and hydrolysis chemistry of the ferritins. Biochim. Biophys. Acta 1800, 719–731 - PubMed
-
- Honarmand Ebrahimi K., Hagedoorn P.-L., and Hagen W. R. (2015) Unity in the biochemistry of the iron-storage proteins ferritin and bacterioferritin. Chem. Rev. 115, 295–326 - PubMed
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

