Mismatch repair deficiency may be common in ductal adenocarcinoma of the prostate
- PMID: 27756888
- PMCID: PMC5347709
- DOI: 10.18632/oncotarget.12697
Mismatch repair deficiency may be common in ductal adenocarcinoma of the prostate
Abstract
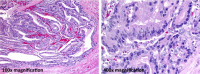
Precision oncology entails making treatment decisions based on a tumor's molecular characteristics. For prostate cancer, identifying clinically relevant molecular subgroups is challenging, as molecular profiling is not routine outside of academic centers. Since histologic variants of other cancers correlates with specific genomic alterations, we sought to determine if ductal adenocarcinoma of the prostate (dPC) - a rare and aggressive histopathologic variant - was associated with any recurrent actionable mutations. Tumors from 10 consecutive patients with known dPC were sequenced on a targeted next-generation DNA sequencing panel. The median age at diagnosis was 59 years (range, 40-73). Four (40%) patients had metastases upon presentation. Archival tissue from formalin-fixed paraffin-embedded prostate tissue samples from nine patients and a biopsy of a metastasis from one patient with castration-resistant prostate cancer were available for analysis. Nine of 10 samples had sufficient material for tumor sequencing. Four (40%) patients' tumors had a mismatch repair (MMR) gene alteration (N = 2, MSH2; N = 1, MSH6; and N = 1, MLH1), of which 3 (75%) had evidence of hypermutation. Sections of the primary carcinomas of three additional patients with known MMR gene alterations/hypermutation were histologically evaluated; two of these tumors had dPC. MMR mutations associated with hypermutation were common in our cohort of dPC patients. Since hypermutation may predict for response to immune checkpoint blockade, the presence of dPC may be a rapid means to enrich populations for further screening. Given our small sample size, these findings require replication.
Keywords: ductal adenocarcinoma; hypermutation; microsatellite instability; mismatch repair; prostate cancer.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
-
- Bessa X, Alenda C, Paya A, Alvarez C, Iglesias M, Seoane A, Dedeu JM, Abuli A, Ilzarbe L, Navarro G, Pellise M, Balaguer F, Castellvi-Bel S, et al. Validation microsatellite path score in a population-based cohort of patients with colorectal cancer. J Clin Oncol. 2011;29:3374–80. doi: 10.1200/jco.2010.34.3947. - DOI - PubMed
-
- Shia J, Ellis NA, Paty PB, Nash GM, Qin J, Offit K, Zhang XM, Markowitz AJ, Nafa K, Guillem JG, Wong WD, Gerald WL, Klimstra DS. Value of histopathology in predicting microsatellite instability in hereditary nonpolyposis colorectal cancer and sporadic colorectal cancer. Am J Surg Pathol. 2003;27:1407–17. - PubMed
-
- Sainty D, Liso V, Cantu-Rajnoldi A, Head D, Mozziconacci MJ, Arnoulet C, Benattar L, Fenu S, Mancini M, Duchayne E, Mahon FX, Gutierrez N, Birg F, et al. A new morphologic classification system for acute promyelocytic leukemia distinguishes cases with underlying PLZF/RARA gene rearrangements. Blood. 2000;96:1287–96. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

