Magnetic Resonance Imaging of Ferumoxytol-Labeled Human Mesenchymal Stem Cells in the Mouse Brain
- PMID: 27757917
- PMCID: PMC5346117
- DOI: 10.1007/s12015-016-9694-0
Magnetic Resonance Imaging of Ferumoxytol-Labeled Human Mesenchymal Stem Cells in the Mouse Brain
Abstract
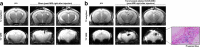
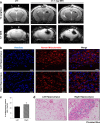
The success of stem cell therapy is highly dependent on accurate delivery of stem cells to the target site of interest. Possible ways to track the distribution of MSCs in vivo include the use of reporter genes or nanoparticles. The U.S. Food and Drug Administration (FDA) has approved ferumoxytol (Feraheme® [USA], Rienso® [UK]) as a treatment for iron deficiency anemia. Ferumoxytol is an ultrasmall superparamagnetic iron oxide nanoparticle (USPIO) that has recently been used to track the fate of transplanted cells using magnetic resonance imaging (MRI). The major objectives of this study were to demonstrate the feasibility of labeling hUCB-MSCs with ferumoxytol and to observe, through MRI, the engraftment of ferumoxytol-labeled human umbilical cord blood-derived mesenchymal stem cells (hUCB-MSCs) delivered via stereotactic injection into the hippocampi of a transgenic mouse model of familial Alzheimer's disease (5XFAD). Ferumoxytol had no toxic effects on the viability or stemness of hUCB-MSCs when assessed in vitro. Through MRI, hypointense signals were discernible at the site where ferumoxytol-labeled human MSCs were injected. Iron-positive areas were also observed in the engrafted hippocampi. The results from this study support the use of nanoparticle labeling to monitor transplanted MSCs in real time as a follow-up for AD stem cell therapy in the clinical field.
Keywords: Ferumoxytol; Magnetic resonance imaging; Mesenchymal stem cells; Monitoring; Stem cell therapy.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures





References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

