Evaluation of solvation free energies for small molecules with the AMOEBA polarizable force field
- PMID: 27757978
- PMCID: PMC5111595
- DOI: 10.1002/jcc.24500
Evaluation of solvation free energies for small molecules with the AMOEBA polarizable force field
Abstract
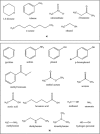
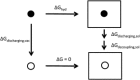
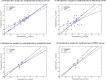
The effects of electronic polarization in biomolecular interactions will differ depending on the local dielectric constant of the environment, such as in solvent, DNA, proteins, and membranes. Here the performance of the AMOEBA polarizable force field is evaluated under nonaqueous conditions by calculating the solvation free energies of small molecules in four common organic solvents. Results are compared with experimental data and equivalent simulations performed with the GAFF pairwise-additive force field. Although AMOEBA results give mean errors close to "chemical accuracy," GAFF performs surprisingly well, with statistically significantly more accurate results than AMOEBA in some solvents. However, for both models, free energies calculated in chloroform show worst agreement to experiment and individual solutes are consistently poor performers, suggesting non-potential-specific errors also contribute to inaccuracy. Scope for the improvement of both potentials remains limited by the lack of high quality experimental data across multiple solvents, particularly those of high dielectric constant. © 2016 The Authors. Journal of Computational Chemistry Published by Wiley Periodicals, Inc.
Keywords: fixed-charge force field; log P; molecular dynamics; polarizable force field; solvation free energies.
© 2016 The Authors. Journal of Computational Chemistry Published by Wiley Periodicals, Inc.
Figures
Similar articles
-
Combining the polarizable Drude force field with a continuum electrostatic Poisson-Boltzmann implicit solvation model.J Comput Chem. 2018 Aug 15;39(22):1707-1719. doi: 10.1002/jcc.25345. Epub 2018 May 8. J Comput Chem. 2018. PMID: 29737546 Free PMC article.
-
Accurate Host-Guest Binding Free Energies Using the AMOEBA Polarizable Force Field.J Chem Inf Model. 2023 May 8;63(9):2769-2782. doi: 10.1021/acs.jcim.3c00155. Epub 2023 Apr 19. J Chem Inf Model. 2023. PMID: 37075788 Free PMC article.
-
Evaluating Parametrization Protocols for Hydration Free Energy Calculations with the AMOEBA Polarizable Force Field.J Chem Theory Comput. 2016 Aug 9;12(8):3871-83. doi: 10.1021/acs.jctc.6b00276. Epub 2016 Jul 29. J Chem Theory Comput. 2016. PMID: 27341007
-
Self-Consistent Reaction Field Model for Aqueous and Nonaqueous Solutions Based on Accurate Polarized Partial Charges.J Chem Theory Comput. 2007 Nov;3(6):2011-33. doi: 10.1021/ct7001418. J Chem Theory Comput. 2007. PMID: 26636198
-
Accounting for electronic polarization in non-polarizable force fields.Phys Chem Chem Phys. 2011 Feb 21;13(7):2613-26. doi: 10.1039/c0cp01971b. Epub 2011 Jan 7. Phys Chem Chem Phys. 2011. PMID: 21212894 Review.
Cited by
-
Force field development phase II: Relaxation of physics-based criteria… or inclusion of more rigorous physics into the representation of molecular energetics.J Comput Aided Mol Des. 2019 Feb;33(2):205-264. doi: 10.1007/s10822-018-0134-x. Epub 2018 Nov 30. J Comput Aided Mol Des. 2019. PMID: 30506159 Review.
-
On the faithfulness of molecular mechanics representations of proteins towards quantum-mechanical energy surfaces.Interface Focus. 2020 Dec 6;10(6):20190121. doi: 10.1098/rsfs.2019.0121. Epub 2020 Oct 16. Interface Focus. 2020. PMID: 33184586 Free PMC article.
-
GAFF/IPolQ-Mod+LJ-Fit: Optimized force field parameters for solvation free energy predictions.ADMET DMPK. 2020 Jun 28;8(3):274-296. doi: 10.5599/admet.837. eCollection 2020. ADMET DMPK. 2020. PMID: 35300308 Free PMC article.
-
Scalable hybrid deep neural networks/polarizable potentials biomolecular simulations including long-range effects.Chem Sci. 2023 Apr 4;14(20):5438-5452. doi: 10.1039/d2sc04815a. eCollection 2023 May 24. Chem Sci. 2023. PMID: 37234902 Free PMC article.
-
A Simple Method for Including Polarization Effects in Solvation Free Energy Calculations When Using Fixed-Charge Force Fields: Alchemically Polarized Charges.ACS Omega. 2020 Jul 7;5(28):17170-17181. doi: 10.1021/acsomega.0c01148. eCollection 2020 Jul 21. ACS Omega. 2020. PMID: 32715202 Free PMC article.
References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

