Extracellular Linkers Completely Transplant the Voltage Dependence from Kv1.2 Ion Channels to Kv2.1
- PMID: 27760355
- PMCID: PMC5072986
- DOI: 10.1016/j.bpj.2016.08.043
Extracellular Linkers Completely Transplant the Voltage Dependence from Kv1.2 Ion Channels to Kv2.1
Abstract
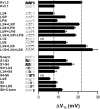
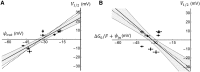
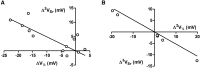
The transmembrane voltage needed to open different voltage-gated K (Kv) channels differs by up to 50 mV from each other. In this study we test the hypothesis that the channels' voltage dependences to a large extent are set by charged amino-acid residues of the extracellular linkers of the Kv channels, which electrostatically affect the charged amino-acid residues of the voltage sensor S4. Extracellular cations shift the conductance-versus-voltage curve, G(V), by interfering with these extracellular charges. We have explored these issues by analyzing the effects of the divalent strontium ion (Sr2+) on the voltage dependence of the G(V) curves of wild-type and chimeric Kv channels expressed in Xenopus oocytes, using the voltage-clamp technique. Out of seven Kv channels, Kv1.2 was found to be most sensitive to Sr2+ (50 mM shifted G(V) by +21.7 mV), and Kv2.1 to be the least sensitive (+7.8 mV). Experiments on 25 chimeras, constructed from Kv1.2 and Kv2.1, showed that the large Sr2+-induced G(V) shift of Kv1.2 can be transferred to Kv2.1 by exchanging the extracellular linker between S3 and S4 (L3/4) in combination with either the extracellular linker between S5 and the pore (L5/P) or that between the pore and S6 (LP/6). The effects of the linker substitutions were nonadditive, suggesting specific structural interactions. The free energy of these interactions was ∼20 kJ/mol, suggesting involvement of hydrophobic interactions and/or hydrogen bonds. Using principles from double-layer theory we derived an approximate linear equation (relating the voltage shifts to altered ionic strength), which proved to well match experimental data, suggesting that Sr2+ acts on these channels mainly by screening surface charges. Taken together, these results highlight the extracellular surface potential at the voltage sensor as an important determinant of the channels' voltage dependence, making the extracellular linkers essential targets for evolutionary selection.
Copyright © 2016. Published by Elsevier Inc.
Figures


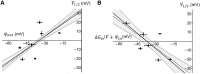



References
-
- Yu F.H., Catterall W.A. The VGL-chanome: a protein superfamily specialized for electrical signaling and ionic homeostasis. Sci. Signal. 2004;2004:re15. - PubMed
-
- Arhem P., Blomberg C. Ion channel density and threshold dynamics of repetitive firing in a cortical neuron model. Biosystems. 2007;89:117–125. - PubMed
-
- Erisir A., Lau D., Leonard C.S. Function of specific K(+) channels in sustained high-frequency firing of fast-spiking neocortical interneurons. J. Neurophysiol. 1999;82:2476–2489. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

