Methamphetamine Induces Anhedonic-Like Behavior and Impairs Frontal Cortical Energetics in Mice
- PMID: 27762079
- PMCID: PMC6492743
- DOI: 10.1111/cns.12649
Methamphetamine Induces Anhedonic-Like Behavior and Impairs Frontal Cortical Energetics in Mice
Abstract
Introduction: We recently showed that a single high dose of methamphetamine (METH) induces a persistent frontal cortical monoamine depletion that is accompanied by helpless-like behavior in mice. However, brain metabolic alterations underlying both neurochemical and mood alterations remain unknown.
Aims: Herein, we aimed at characterizing frontal cortical metabolic alterations associated with early negative mood behavior triggered by METH. Adult C57BL/6 mice were injected with METH (30 mg/kg, i.p.), and their frontal cortical metabolic status was characterized after probing their mood and anxiety-related phenotypes 3 days postinjection.
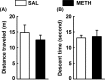
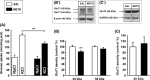
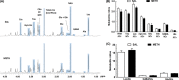
Results: Methamphetamine induced depressive-like behavior, as indicated by the decreased grooming time in the splash test and by a transient decrease in sucrose preference. At this time, METH did not alter anxiety-like behavior or motor functions. Depolarization-induced glucose uptake was reduced in frontocortical slices from METH-treated mice compared to controls. Consistently, astrocytic glucose transporter (GluT1) density was lower in the METH group. A proton high rotation magic angle spinning (HRMAS) spectroscopic approach revealed that METH induced a significant decrease in N-acetyl aspartate (NAA) and glutamate levels, suggesting that METH decreased neuronal glutamatergic function in frontal cortex.
Conclusions: We report, for the first time, that a single METH injection triggers early self-care and hedonic deficits and impairs frontal cortical energetics in mice.
Keywords: Depressive-like behavior; Frontal cortex; Glucose metabolism; Methamphetamine.
© 2016 John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Cruicshank CC, Dyer KR. A review of the clinical pharmacology of methamphetamine. Addiction 2009;104:1085–1099. - PubMed
-
- McGregor C, Srisurapanont M, Jittiwutikarn J, Laobhripatr S, Wongtan T, White JM. The nature, time course and severity of methamphetamine withdrawal. Addiction 2005;100:1320–1329. - PubMed
-
- Silva CD, Neves AF, Dias AI, et al. A single neurotoxic dose of methamphetamine induces a long‐lasting depressive‐like behavior in mice. Neurotox Res 2014;25:295–304. - PubMed
-
- London ED, Simon SL, Berman SM, et al. Mood disturbances and regional cerebral metabolic abnormalities in recently abstinent methamphetamine abusers. Arch Gen Psychiatry 2004;61:73–84. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

