Microtubule Organization Determines Axonal Transport Dynamics
- PMID: 27764672
- PMCID: PMC5432135
- DOI: 10.1016/j.neuron.2016.09.036
Microtubule Organization Determines Axonal Transport Dynamics
Abstract
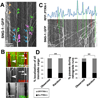
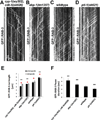
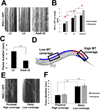
Axonal microtubule (MT) arrays are the major cytoskeleton substrate for cargo transport. How MT organization, i.e., polymer length, number, and minus-end spacing, is regulated and how it impinges on axonal transport are unclear. We describe a method for analyzing neuronal MT organization using light microscopy. This method circumvents the need for electron microscopy reconstructions and is compatible with live imaging of cargo transport and MT dynamics. Examination of a C. elegans motor neuron revealed how age, MT-associated proteins, and signaling pathways control MT length, minus-end spacing, and coverage. In turn, MT organization determines axonal transport progression: cargoes pause at polymer termini, suggesting that switching MT tracks is rate limiting for efficient transport. Cargo run length is set by MT length, and higher MT coverage correlates with shorter pauses. These results uncover the principles and mechanisms of neuronal MT organization and its regulation of axonal cargo transport.
Keywords: C. elegans; axonal transport; dynein; kinesin; microtubule; microtubule dynamics; microtubule length; microtubule organization.
Copyright © 2016 Elsevier Inc. All rights reserved.
Figures



References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

