Single-Flask Multicomponent Synthesis of Highly Substituted α-Pyrones via a Sequential Enolate Arylation and Alkenylation Strategy
- PMID: 27768319
- PMCID: PMC8103787
- DOI: 10.1021/acs.orglett.6b02969
Single-Flask Multicomponent Synthesis of Highly Substituted α-Pyrones via a Sequential Enolate Arylation and Alkenylation Strategy
Abstract
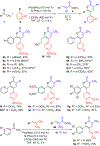
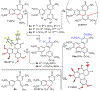
Trisubstituted α-pyrones are obtained by a Pd-catalyzed three-component, single-flask operation via an α-arylation, subsequent α-alkenylation, alkene isomerization, and dienolate lactonization. A variety of coupling components under mild conditions afforded isolated yields of up to 93% of the pyrones with complete control of regioselectivity. Metal dependence was noted for three of the steps of the pathway. Utility of the pyrone products was demonstrated by further transformations providing convenient access to polyaromatic compounds, exhibiting broad molecular diversity.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
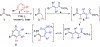

Similar articles
-
Palladium-catalyzed difunctionalization of internal alkynes via highly regioselective 6-endo cyclization and alkenylation of enynoates: synthesis of multisubstituted pyrones.Org Lett. 2015 Apr 3;17(7):1636-9. doi: 10.1021/acs.orglett.5b00272. Epub 2015 Mar 19. Org Lett. 2015. PMID: 25789819
-
Recent advances in efficient and selective synthesis of di-, tri-, and tetrasubstituted alkenes via Pd-catalyzed alkenylation-carbonyl olefination synergy.Acc Chem Res. 2008 Nov 18;41(11):1474-85. doi: 10.1021/ar800038e. Acc Chem Res. 2008. PMID: 18783256
-
Convergent synthesis of fostriecin via selective alkene couplings and regioselective asymmetric dihydroxylation.Org Lett. 2009 Dec 3;11(23):5498-501. doi: 10.1021/ol902365n. Org Lett. 2009. PMID: 19902969
-
From α-arylation of olefins to acylation with aldehydes: a journey in regiocontrol of the Heck reaction.Acc Chem Res. 2011 Aug 16;44(8):614-26. doi: 10.1021/ar200053d. Epub 2011 May 25. Acc Chem Res. 2011. PMID: 21612205 Review.
-
Enantioselective Dicarbofunctionalization of Unactivated Alkenes by Palladium-Catalyzed Tandem Heck/Suzuki Coupling Reaction.Angew Chem Int Ed Engl. 2019 Oct 7;58(41):14653-14659. doi: 10.1002/anie.201907840. Epub 2019 Sep 5. Angew Chem Int Ed Engl. 2019. PMID: 31420928 Review.
Cited by
-
Diversification reactions of γ-silyl allenyl esters: selective conversion to all-carbon quaternary centers and γ-allene dicarbinols.Chem Commun (Camb). 2017 May 4;53(37):5125-5127. doi: 10.1039/c7cc01708a. Chem Commun (Camb). 2017. PMID: 28435948 Free PMC article.
References
-
- Afarinkia K; Vinader V; Nelson TD; Posner GH Tetrahedron 1992, 48, 9111.
- Sun C-L; Fürstner A Angew. Chem., Int. Ed 2013, 52, 13071. - PubMed
- Shin H-S; Jung Y-G; Cho H-K; Park H-K; Cho C-G Org. Lett 2014, 16, 5718. - PubMed
- Okura K; Tamura R; Shigehara K; Masai E; Nakamura M; Otsuka Y; Katayama Y; Nakao Y Chem. Lett 2014, 43, 1349.
- Khatri AI; Samant SD RSC Adv 2015, 5, 2009.
- Gan P; Smith MW; Braffman NR; Snyder SA Angew. Chem., Int. Ed 2016, 55, 3625. - PubMed
-
- Goel A; Taneja G; Raghuvanshi A; Kant R; Maulik PR Org. Biomol. Chem 2013, 11, 5239. - PubMed
- Miura T; Fujioka S; Takemura N; Iwasaki H; Ozeki M; Kojima N; Yamashita M Synthesis 2014, 46, 496.
- Yeh P-P; Daniels DSB; Cordes DB; Slawin AMZ; Smith AD Org. Lett 2014, 16, 964. - PubMed
- Liu W; Zhao G Org. Biomol. Chem 2014, 12, 832. - PubMed
-
- Mochida S; Hirano K; Satoh T; Miura MJ Org. Chem 2009, 74, 6295. - PubMed
- Ackermann L; Pospech J; Graczyk K; Rauch K Org. Lett 2012, 14, 930. - PubMed
- Li Q; Yan Y; Wang X; Gong B; Tang X; Shi J; Xu HE; Yi W RSC Adv 2013, 3, 23402.
- Yu Y; Huang L; Wu W; Jiang H Org. Lett 2014, 16, 2146. - PubMed
- Moghaddam FM; Mirjafary Z; Javan MJ; Motamen S; Saeidian H Tetrahedron Lett 2014, 55, 2908.
- Manikandan R; Jeganmohan M Org. Lett 2014, 16, 652. - PubMed
- Tian P-P; Cai S-H; Liang Q-J; Zhou X-Y; Xu Y-H; Loh T-P Org. Lett 2015, 17, 1636. - PubMed
- Preindl J; Jouvin K; Laurich D; Seidel G; Fürstner A Chem. -Eur. J 2016, 22, 237. - PubMed
- Faizi DJ; Issaian A; Davis AJ; Blum SA J. Am. Chem. Soc 2016, 138, 2126. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

