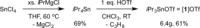Versatile Catalytic Hydrogenation Using A Simple Tin(IV) Lewis Acid
- PMID: 27774711
- PMCID: PMC5129554
- DOI: 10.1002/anie.201606639
Versatile Catalytic Hydrogenation Using A Simple Tin(IV) Lewis Acid
Abstract
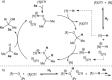
Despite the rapid development of frustrated Lewis pair (FLP) chemistry over the last ten years, its application in catalytic hydrogenations remains dependent on a narrow family of structurally similar early main-group Lewis acids (LAs), inevitably placing limitations on reactivity, sensitivity and substrate scope. Herein we describe the FLP-mediated H2 activation and catalytic hydrogenation activity of the alternative LA iPr3 SnOTf, which acts as a surrogate for the trialkylstannylium ion iPr3 Sn+ , and is rapidly and easily prepared from simple, inexpensive starting materials. This highly thermally robust LA is found to be competent in the hydrogenation of a number of different unsaturated functional groups (which is unique to date for main-group FLP LAs not based on boron), and also displays a remarkable tolerance to moisture.
Keywords: catalysis; frustrated Lewis pairs; hydrogenation; stannylium; tin.
© 2016 The Authors. Published by Wiley-VCH Verlag GmbH & Co. KGaA.
Figures
Similar articles
-
Hydrogen activation using a novel tribenzyltin Lewis acid.Philos Trans A Math Phys Eng Sci. 2017 Aug 28;375(2101):20170008. doi: 10.1098/rsta.2017.0008. Philos Trans A Math Phys Eng Sci. 2017. PMID: 28739966 Free PMC article.
-
Mechanistic Insights into Hydrogen Activation by Frustrated N/Sn Lewis Pairs.Chemistry. 2018 Feb 21;24(11):2575-2579. doi: 10.1002/chem.201705861. Epub 2018 Jan 25. Chemistry. 2018. PMID: 29276811
-
Catalytic hydrogenation with frustrated Lewis pairs: selectivity achieved by size-exclusion design of Lewis acids.Chemistry. 2012 Jan 9;18(2):574-85. doi: 10.1002/chem.201102438. Epub 2011 Dec 12. Chemistry. 2012. PMID: 22161804
-
Frustrated Lewis Pairs: Bonding, Reactivity, and Applications.J Phys Chem A. 2023 Jun 1;127(21):4561-4582. doi: 10.1021/acs.jpca.3c02141. Epub 2023 May 22. J Phys Chem A. 2023. PMID: 37216335 Review.
-
FLP catalysis: main group hydrogenations of organic unsaturated substrates.Chem Soc Rev. 2019 Jul 1;48(13):3592-3612. doi: 10.1039/c8cs00277k. Chem Soc Rev. 2019. PMID: 30178796 Review.
Cited by
-
Hydrogen activation using a novel tribenzyltin Lewis acid.Philos Trans A Math Phys Eng Sci. 2017 Aug 28;375(2101):20170008. doi: 10.1098/rsta.2017.0008. Philos Trans A Math Phys Eng Sci. 2017. PMID: 28739966 Free PMC article.
-
Metal-Ligand Cooperation Facilitates Bond Activation and Catalytic Hydrogenation with Zinc Pincer Complexes.J Am Chem Soc. 2020 Aug 26;142(34):14513-14521. doi: 10.1021/jacs.0c05500. Epub 2020 Aug 17. J Am Chem Soc. 2020. PMID: 32786799 Free PMC article.
-
Trioxatriangulenium (TOTA+) as a robust carbon-based Lewis acid in frustrated Lewis pair chemistry.Chem Sci. 2021 Feb 8;12(13):4841-4849. doi: 10.1039/d0sc05893a. Chem Sci. 2021. PMID: 34168760 Free PMC article.
-
Differences in the Reactivity of Geminal Si-O-P and Al-O-P Frustrated Lewis Pairs.Chemistry. 2023 Feb 7;29(8):e202202842. doi: 10.1002/chem.202202842. Epub 2022 Dec 19. Chemistry. 2023. PMID: 36349870 Free PMC article.
-
A Zwitterionic Phosphonium Stannate(II) via Hydrogen Splitting by a Sn/P Frustrated Lewis-Pair and Reductive Elimination.Chemistry. 2020 Dec 23;26(72):17381-17385. doi: 10.1002/chem.202004425. Epub 2020 Nov 23. Chemistry. 2020. PMID: 33016507 Free PMC article.
References
-
- For key publications, see:
-
- Welch G. C., San Juan R. R., Masuda J. D., Stephan D. W., Science 2006, 314, 1124–1126; - PubMed
-
- Welch G. C., Stephan D. W., J. Am. Chem. Soc. 2007, 129, 1880–1881; - PubMed
-
- Chase P. A., Welch G. C., Jurca T., Stephan D. W., Angew. Chem. Int. Ed. 2007, 46, 8050–8053; - PubMed
- Angew. Chem. 2007, 119, 8196–8199;
-
- Chase P. A., Jurca T., Stephan D. W., Chem. Commun. 2008, 1701–1703; - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources