Differential response to hypomethylating agents based on sex: a report on behalf of the MDS Clinical Research Consortium (MDS CRC)
- PMID: 27774847
- PMCID: PMC5394924
- DOI: 10.1080/10428194.2016.1246726
Differential response to hypomethylating agents based on sex: a report on behalf of the MDS Clinical Research Consortium (MDS CRC)
Abstract
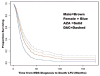
First-line therapy for higher-risk myelodysplastic syndromes (MDS) includes decitabine (DAC) or azacitidine (AZA). Variables have not identified differential response rates between these. We assessed the influence of patient sex on outcomes including overall survival (OS) in 642 patients with higher-risk MDS treated with AZA or DAC. DAC-treated patients (35% of females, 31% of males) had marginally better OS than AZA-treated patients (p = .043), (median OS of 18.7 months versus 16.4 months), but the difference varied strongly by sex. Female patients treated with DAC had a longer median OS (21.1 months, 95% CI: 16.0-28.0) than female patients treated with AZA (13.2 months, 95% CI: 11.0-15.9; p = .0014), while for males there was no significant difference between HMAs (median OS 18.3 months with DAC versus 17.9 months for AZA, p = .59). The biological reason for this variability is unclear, but may be a consequence of differences in cytidine deaminase activity between men and women.
Keywords: MDS; Sex; azacitidine; decitabine; hypomethylating agents.
Conflict of interest statement
Figures
Comment in
-
Choosing a hypomethylating agent in MDS: does gender matter?Leuk Lymphoma. 2017 Jun;58(6):1277-1278. doi: 10.1080/10428194.2017.1283035. Epub 2017 Jan 31. Leuk Lymphoma. 2017. PMID: 28140711 No abstract available.
Similar articles
-
Elevated fetal haemoglobin is a predictor of better outcome in MDS/AML patients receiving 5-aza-2'-deoxycytidine (Decitabine).Br J Haematol. 2017 Feb;176(4):609-617. doi: 10.1111/bjh.14463. Epub 2016 Dec 1. Br J Haematol. 2017. PMID: 27905102 Clinical Trial.
-
Comparison of Azacitidine and Decitabine in Myelodysplastic Syndromes and Acute Myeloid Leukemia: A Network Meta-analysis.Clin Lymphoma Myeloma Leuk. 2021 Jun;21(6):e530-e544. doi: 10.1016/j.clml.2021.01.024. Epub 2021 Feb 24. Clin Lymphoma Myeloma Leuk. 2021. PMID: 33716056
-
Treatment of relapsed AML and MDS after allogeneic stem cell transplantation with decitabine and DLI-a retrospective multicenter analysis on behalf of the German Cooperative Transplant Study Group.Ann Hematol. 2018 Feb;97(2):335-342. doi: 10.1007/s00277-017-3185-5. Epub 2017 Nov 18. Ann Hematol. 2018. PMID: 29151133
-
Digging deep into "dirty" drugs - modulation of the methylation machinery.Drug Metab Rev. 2015 May;47(2):252-79. doi: 10.3109/03602532.2014.995379. Epub 2015 Jan 8. Drug Metab Rev. 2015. PMID: 25566693 Free PMC article. Review.
-
The euphoria of hypomethylating agents in MDS and AML: is it justified?Best Pract Res Clin Haematol. 2013 Sep;26(3):275-8. doi: 10.1016/j.beha.2013.10.001. Epub 2013 Oct 15. Best Pract Res Clin Haematol. 2013. PMID: 24309530 Review.
Cited by
-
Acute myeloid leukemia: does sex matter?Leukemia. 2024 Nov;38(11):2329-2331. doi: 10.1038/s41375-024-02435-z. Epub 2024 Oct 14. Leukemia. 2024. PMID: 39402216 Free PMC article. Review. No abstract available.
-
Sex Disparities in Myelodysplastic Syndromes: Genotype, Phenotype, and Outcomes.Clin Lymphoma Myeloma Leuk. 2023 May;23(5):355-359. doi: 10.1016/j.clml.2023.01.007. Epub 2023 Jan 16. Clin Lymphoma Myeloma Leuk. 2023. PMID: 36813626 Free PMC article.
-
Decitabine- and 5-azacytidine resistance emerges from adaptive responses of the pyrimidine metabolism network.Leukemia. 2021 Apr;35(4):1023-1036. doi: 10.1038/s41375-020-1003-x. Epub 2020 Aug 7. Leukemia. 2021. PMID: 32770088 Free PMC article.
-
How I Manage Transplant Ineligible Patients with Myelodysplastic Neoplasms.Clin Hematol Int. 2023 Mar;5(1):8-20. doi: 10.1007/s44228-022-00024-4. Epub 2022 Dec 27. Clin Hematol Int. 2023. PMID: 36574201 Free PMC article. Review.
-
Sex Differences in the Spectrum of Clonal Hematopoiesis.Hemasphere. 2023 Jan 24;7(2):e832. doi: 10.1097/HS9.0000000000000832. eCollection 2023 Feb. Hemasphere. 2023. PMID: 36713353 Free PMC article. No abstract available.
References
-
- Vardiman JW, Thiele J, Arber DA, et al. The 2008 revision of the World Health Organization (WHO) classification of myeloid neoplasms and acute leukemia: rationale and important changes. Blood. 2009;114:937–951. - PubMed
-
- Cogle CR, Iannacone MR, Yu D, et al. High rate of uncaptured myelodysplastic syndrome cases and an improved method of case ascertainment. Leuk Res. 2014;38:71–75. - PubMed
-
- Sekeres MA. Epidemiology, natural history, and practice patterns of patients with myelodysplastic syndromes in 2010. JNCCN. 2011;9:57–63. - PubMed
-
- Sekeres MA. The myelodysplastic syndromes. Exp Opin Biol Ther. 2007;7:369–377. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous