A novel ligand of calcitonin receptor reveals a potential new sensor that modulates programmed cell death
- PMID: 27777788
- PMCID: PMC5056446
- DOI: 10.1038/cddiscovery.2016.62
A novel ligand of calcitonin receptor reveals a potential new sensor that modulates programmed cell death
Abstract
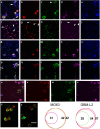
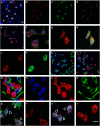
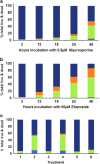
We have discovered that the accumulation of an anti-calcitonin receptor (anti-CTR) antibody conjugated to a fluorophore (mAb2C4:AF568) provides a robust signal for cells undergoing apoptotic programmed cell death (PCD). PCD is an absolute requirement for normal development of metazoan organisms. PCD is a hallmark of common diseases such as cardiovascular disease and tissue rejection in graft versus host pathologies, and chemotherapeutics work by increasing PCD. This robust signal or high fluorescent events were verified by confocal microscopy and flow cytometry in several cell lines and a primary culture in which PCD had been induced. In Jurkat cells, GBM-L2 and MG63 cells, the percentage undergoing PCD that were positive for both mAb2C4:AF568 and annexin V ranged between 70 and >90%. In MG63 cells induced for the preapoptotic cell stress response (PACSR), the normal expression of α-tubulin, a key structural component of the cytoskeleton, and accumulation of mAb2C4:AF568 were mutually exclusive. Our data support a model in which CTR is upregulated during PACSR and recycles to the plasma membrane with apoptosis. In cells committed to apoptosis (α-tubulin negative), there is accumulation of the CTR-ligand mAb2C4:AF568 generating a high fluorescent event. The reagent mAb2C4:AF568 effectively identifies a novel event linked to apoptosis.
Conflict of interest statement
PJW and SGBF are named inventors on the Int PCT AU2014/001081, filed on 28 November 2014. PJW is a director of Welcome Receptor Antibodies Pty Ltd (Australia), which filed the patent application. The other authors declare no conflict of interest.
Figures

Similar articles
-
Dianthin-30 or gelonin versus monomethyl auristatin E, each configured with an anti-calcitonin receptor antibody, are differentially potent in vitro in high-grade glioma cell lines derived from glioblastoma.Cancer Immunol Immunother. 2017 Sep;66(9):1217-1228. doi: 10.1007/s00262-017-2013-z. Epub 2017 May 13. Cancer Immunol Immunother. 2017. PMID: 28501939 Free PMC article.
-
Programmed cell death (PCD). Apoptosis, autophagic PCD, or others?Ann N Y Acad Sci. 2000;926:1-12. doi: 10.1111/j.1749-6632.2000.tb05594.x. Ann N Y Acad Sci. 2000. PMID: 11193023 Review.
-
Cell-surface exposure of phosphatidylserine correlates with the stage of fludarabine-induced apoptosis in chronic lymphocytic leukemia and expression of apoptosis-regulating genes.Cytometry. 2000 May 1;40(1):19-25. doi: 10.1002/(sici)1097-0320(20000501)40:1<19::aid-cyto3>3.0.co;2-3. Cytometry. 2000. PMID: 10754513
-
Sequestration of mitotic (M-phase) chromosomes in autophagosomes: mitotic programmed cell death in human Chang liver cells induced by an OH* burst from vanadyl(4).Anat Rec. 1996 May;245(1):1-8. doi: 10.1002/(SICI)1097-0185(199605)245:1<1::AID-AR1>3.0.CO;2-2. Anat Rec. 1996. PMID: 8731032
-
Initiation of programmed cell death in self-incompatibility: role for cytoskeleton modifications and several caspase-like activities.Mol Plant. 2008 Nov;1(6):879-87. doi: 10.1093/mp/ssn053. Epub 2008 Oct 17. Mol Plant. 2008. PMID: 19825589 Review.
Cited by
-
Expression and activity of the calcitonin receptor family in a sample of primary human high-grade gliomas.BMC Cancer. 2019 Feb 18;19(1):157. doi: 10.1186/s12885-019-5369-y. BMC Cancer. 2019. PMID: 30777055 Free PMC article.
-
Building the case for the calcitonin receptor as a viable target for the treatment of glioblastoma.Ther Adv Med Oncol. 2020 Dec 18;12:1758835920978110. doi: 10.1177/1758835920978110. eCollection 2020. Ther Adv Med Oncol. 2020. PMID: 33425026 Free PMC article. Review.
-
Dianthin-30 or gelonin versus monomethyl auristatin E, each configured with an anti-calcitonin receptor antibody, are differentially potent in vitro in high-grade glioma cell lines derived from glioblastoma.Cancer Immunol Immunother. 2017 Sep;66(9):1217-1228. doi: 10.1007/s00262-017-2013-z. Epub 2017 May 13. Cancer Immunol Immunother. 2017. PMID: 28501939 Free PMC article.
-
CGRP and the Calcitonin Receptor are Co-Expressed in Mouse, Rat and Human Trigeminal Ganglia Neurons.Front Physiol. 2022 May 10;13:860037. doi: 10.3389/fphys.2022.860037. eCollection 2022. Front Physiol. 2022. PMID: 35620595 Free PMC article.
-
Methods to measure calcitonin receptor activity, up-regulated in cell stress, apoptosis and autophagy.F1000Res. 2021 Oct 7;10:1019. doi: 10.12688/f1000research.72845.1. eCollection 2021. F1000Res. 2021. PMID: 34745567 Free PMC article. Review.
References
-
- Vanden Berghe T, Linkermann A, Jouan-Lanhouet S, Walczak H, Vandenabeele P. Regulated necrosis: the expanding network of non-apoptotic cell death pathways. Nat Rev Mol Cell Biol 2014; 15: 135–147. - PubMed
-
- Pasparakis M, Vandenabeele P. Necroptosis and its role in inflammation. Nature 2015; 517: 311–320. - PubMed
-
- Lalaoui N, Lindqvist LM, Sandow JJ, Ekert PG. The molecular relationships between apoptosis, autophagy and necroptosis. Semin Cell Dev Biol 2015; 39: 63–69. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

