The transfer of multigene panel testing for hereditary breast and ovarian cancer to healthcare: What are the implications for the management of patients and families?
- PMID: 27779110
- PMCID: PMC5356770
- DOI: 10.18632/oncotarget.12699
The transfer of multigene panel testing for hereditary breast and ovarian cancer to healthcare: What are the implications for the management of patients and families?
Abstract
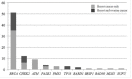
Until recently, the molecular diagnosis of hereditary breast and ovarian cancer (HBOC) was mostly based on BRCA1/2 testing. Next generation sequencing and the recent discovery of new genes involved in HBOC now permit the transfer of genomic capture targeting multiple candidate genes from research to clinical use. However, the implications for the management of patients and their families have not been extensively studied, in particular since some of these genes are not well-established cancer predisposing genes. We studied 583 consecutive patients from Burgundy (France) fulfilling the criteria for BRCA testing using a next generation sequencing 25-genes panel including 20 well-established high-risk cancer genes as well as more recently identified predisposing HBOC cancer. A pathogenic BRCA1/2 mutation was found in 51 patients (9%). Besides, we found 37 pathogenic or likely pathogenic mutations in 10 different high to low-risk genes in 34 patients (6%). The most frequently mutated genes were CHEK2 (n = 12; 2%), ATM (n = 9; 1.5%), and PALB2 (n = 4; 0.6%). Three patients had a mutation in two different predisposing genes. The analysis of clinical actionability conducted in mutation-positive individuals revealed that additional disease-specific screening and/or prevention measures beyond those based on personal and family history alone had been recommended in 69% of cases. In conclusion, multigene panel testing is a powerful tool to identifying high to low-risk HBOC susceptibility genes. The penetrance and spectrum of cancers with these other genes are sometimes undefined, and further collaborative work is crucial to address this question.
Keywords: breast and ovarian cancer susceptibility genes; candidate genes; genomic capture; management; next generation sequencing.
Conflict of interest statement
The authors report no conflict of interest
Figures


References
-
- Weissman SW, Weiss SM, Newlin AC. Genetic testing by cancer site ovary. Cancer J. 2012;18:320–7. - PubMed
-
- Antoniou AC, Pharoah PDP, Narod S, Risch HA, Eyfjord JE, Hopper JL, Loman N, Olsson H, Johannsson O, Borg A, Pasini B, Radice P, Manoukian S, et al. Average risks of breast and ovarian cancer associated with BRCA1 or BRCA2 mutations detected in case series unselected for family history: a combined analysis of 22 studies. Am J Hum Genet. 2003;72:1117–1130. - PMC - PubMed
-
- Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013;63:11–30. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

