Yellow fever vaccine-associated viscerotropic disease: current perspectives
- PMID: 27784992
- PMCID: PMC5066857
- DOI: 10.2147/DDDT.S99600
Yellow fever vaccine-associated viscerotropic disease: current perspectives
Abstract
Purpose: To assess those published cases of yellow fever (YF) vaccine-associated viscerotropic disease that meet the Brighton Collaboration criteria and to assess the safety of YF vaccine with respect to viscerotropic disease.
Literature search: Ten electronic databases were searched with no restriction of date or language and reference lists of retrieved articles.
Methods: All abstracts and titles were independently read by two reviewers and data independently entered by two reviewers.
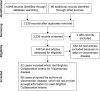
Results: All serious adverse events that met the Brighton Classification criteria were associated with first YF vaccinations. Sixty-two published cases (35 died) met the Brighton Collaboration viscerotropic criteria, with 32 from the US, six from Brazil, five from Peru, three from Spain, two from the People's Republic of China, one each from Argentina, Australia, Belgium, Ecuador, France, Germany, Ireland, New Zealand, Portugal, and the UK, and four with no country stated. Two cases met both the viscerotropic and YF vaccine-associated neurologic disease criteria. Seventy cases proposed by authors as viscerotropic disease did not meet any Brighton Collaboration viscerotropic level of diagnostic certainty or any YF vaccine-associated viscerotropic disease causality criteria (37 died).
Conclusion: Viscerotropic disease is rare in the published literature and in pharmacovigilance databases. All published cases were from developing countries. Because the symptoms are usually very severe and life threatening, it is unlikely that cases would not come to medical attention (but might not be published). Because viscerotropic disease has a highly predictable pathologic course, it is likely that viscerotropic disease post-YF vaccine occurs in low-income countries with the same incidence as in developing countries. YF vaccine is a very safe vaccine that likely confers lifelong immunity.
Keywords: postvaccination severe adverse events; systematic review; viscerotropic disease; yellow fever vaccine.
Conflict of interest statement
The author reports no conflicts of interest in this work.
Figures
Similar articles
-
How many published cases of serious adverse events after yellow fever vaccination meet Brighton Collaboration diagnostic criteria?Vaccine. 2013 Dec 16;31(52):6201-9. doi: 10.1016/j.vaccine.2013.10.050. Vaccine. 2013. PMID: 24436960
-
Active and passive surveillance of yellow fever vaccine 17D or 17DD-associated serious adverse events: systematic review.Vaccine. 2011 Jun 20;29(28):4544-55. doi: 10.1016/j.vaccine.2011.04.055. Epub 2011 May 5. Vaccine. 2011. PMID: 21549787
-
Risk of yellow fever vaccine-associated viscerotropic disease among the elderly: a systematic review.Vaccine. 2013 Dec 2;31(49):5798-805. doi: 10.1016/j.vaccine.2013.09.030. Epub 2013 Sep 27. Vaccine. 2013. PMID: 24079979
-
A rapid and systematic review of the clinical effectiveness and cost-effectiveness of topotecan for ovarian cancer.Health Technol Assess. 2001;5(28):1-110. doi: 10.3310/hta5280. Health Technol Assess. 2001. PMID: 11701100
-
Interventions targeted at women to encourage the uptake of cervical screening.Cochrane Database Syst Rev. 2021 Sep 6;9(9):CD002834. doi: 10.1002/14651858.CD002834.pub3. Cochrane Database Syst Rev. 2021. PMID: 34694000 Free PMC article.
Cited by
-
Recurrent pityriasis rosea: A case report.Hum Vaccin Immunother. 2018 Apr 3;14(4):1024-1026. doi: 10.1080/21645515.2017.1409928. Epub 2017 Dec 21. Hum Vaccin Immunother. 2018. PMID: 29182459 Free PMC article.
-
Adverse events following immunization (AEFI) with fractional one-fifth and one-half doses of yellow fever vaccine compared to full dose in children 9-23 months old in Uganda, 2019-2020 - Preliminary report.Vaccine. 2024 Sep 17;42(22):126197. doi: 10.1016/j.vaccine.2024.126197. Epub 2024 Aug 15. Vaccine. 2024. PMID: 39153293 Free PMC article. Clinical Trial.
-
What Does the Future Hold for Yellow Fever Virus? (II).Genes (Basel). 2018 Aug 21;9(9):425. doi: 10.3390/genes9090425. Genes (Basel). 2018. PMID: 30134625 Free PMC article. Review.
-
Historical Perspectives on Flavivirus Research.Viruses. 2017 Apr 30;9(5):97. doi: 10.3390/v9050097. Viruses. 2017. PMID: 28468299 Free PMC article. Review.
-
Severe Multiorgan Failure Following Yellow Fever Vaccination.Vaccines (Basel). 2020 May 26;8(2):249. doi: 10.3390/vaccines8020249. Vaccines (Basel). 2020. PMID: 32466490 Free PMC article.
References
-
- Monath TP, Vasconcelos PF. Yellow fever. J Clin Virol. 2015;64:160–173. - PubMed
-
- Quaresma JAS, Pagliari C, Medeiros DBA, Duarte MIS, Vasconcelos PFC. Immunity and immune response, pathology and pathologic changes: progress and challenges in the immunopathology of yellow fever. Rev Med Virol. 2013;23(5):305–318. - PubMed
-
- Jentes ES, Poumerol G, Gershman MD, et al. Informal WHO Working Group on Geographic Risk for Yellow Fever The revised global yellow fever risk map and recommendations for vaccination, 2010: consensus of the Informal WHO Working Group on Geographic Risk for Yellow Fever. Lancet Infect Dis. 2011;11(8):622–632. - PubMed
-
- Hill DR. Mapping the risk of yellow fever infection. Curr Infect Dis Rep. 2012;14(3):246–255. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous