Evaluating frequency and quality of pathogen-specific T cells
- PMID: 27786275
- PMCID: PMC5095286
- DOI: 10.1038/ncomms13264
Evaluating frequency and quality of pathogen-specific T cells
Abstract
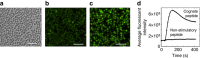
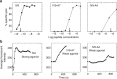
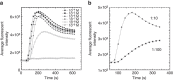
It is generally accepted that enumeration and characterization of antigen-specific T cells provide essential information about potency of the immune response. Here, we report a new technique to determine the frequency and potency of antigen-specific CD8 T cells. The assay measures changes of intracellular Ca2+ in real time by fluorescent microscopy in individual CD8 T cells responding to cognate peptides. The T cells form continuous monolayer, enabling the cells to present the peptides to each other. This approach allows us to evaluate the kinetics of intracellular Ca2+ signalling that characterizes the quality of T cell response. We demonstrate the usefulness of the assay examining the frequency and quality of cytomegalovirus-specific CD8 T cells from healthy donor and patient after haploidentical stem cell transplantation. The new assay has a potential to provide essential information determining the status of the immune system, disease morbidity, potency of therapeutic intervention and vaccine efficacy.
Conflict of interest statement
The assay is protected by pending patent (US62/183,997), inventors Y.S., N.A., N.F. and D.G. The remaining authors declare no competing financial interests.
Figures


Similar articles
-
Reconstitution of cytomegalovirus-specific T-cell response in allogeneic hematopoietic stem cell recipients: the contribution of six frequently recognized, virus-encoded ORFs.Transpl Infect Dis. 2016 Jun;18(3):381-9. doi: 10.1111/tid.12540. Epub 2016 Jun 9. Transpl Infect Dis. 2016. PMID: 27061389
-
Normalizing ELISPOT responses to T-cell counts: a novel approach for quantification of HCMV-specific CD4(+) and CD8(+) T-cell responses in kidney transplant recipients.J Clin Virol. 2014 Sep;61(1):65-73. doi: 10.1016/j.jcv.2014.05.017. Epub 2014 Jun 4. J Clin Virol. 2014. PMID: 24961915
-
Diversified phenotype of antigen specific CD8+ T cells responding to the immunodominant epitopes of IE and pp65 antigens of human cytomegalovirus.Cell Immunol. 2015 Jun;295(2):105-11. doi: 10.1016/j.cellimm.2015.03.008. Epub 2015 Mar 30. Cell Immunol. 2015. PMID: 25880101
-
Evaluation of human cytomegalovirus-specific CD8+ T-cells in allogeneic haematopoietic stem cell transplant recipients using pentamer and interferon-γ-enzyme-linked immunospot assays.J Clin Virol. 2013 Oct;58(2):427-31. doi: 10.1016/j.jcv.2013.07.006. Epub 2013 Jul 30. J Clin Virol. 2013. PMID: 23910932
-
Molecular characterization of HCMV-specific immune responses: Parallels between CD8(+) T cells, CD4(+) T cells, and NK cells.Eur J Immunol. 2015 Sep;45(9):2433-45. doi: 10.1002/eji.201545495. Epub 2015 Aug 24. Eur J Immunol. 2015. PMID: 26228786 Review.
Cited by
-
The Presence of a CMV Immunodominant Allele in the Recipient Is Associated With Increased Survival in CMV Positive Patients Undergoing Haploidentical Hematopoietic Stem Cell Transplantation.Front Oncol. 2019 Sep 17;9:888. doi: 10.3389/fonc.2019.00888. eCollection 2019. Front Oncol. 2019. PMID: 31608225 Free PMC article.
-
Microfluidic T Cell Selection by Cellular Avidity.Adv Healthc Mater. 2022 Aug;11(16):e2200169. doi: 10.1002/adhm.202200169. Epub 2022 Jun 21. Adv Healthc Mater. 2022. PMID: 35657072 Free PMC article.
-
Novel Identified HLA-A*0201-Restricted Hantaan Virus Glycoprotein Cytotoxic T-Cell Epitopes Could Effectively Induce Protective Responses in HLA-A2.1/Kb Transgenic Mice May Associate with the Severity of Hemorrhagic Fever with Renal Syndrome.Front Immunol. 2017 Dec 12;8:1797. doi: 10.3389/fimmu.2017.01797. eCollection 2017. Front Immunol. 2017. PMID: 29312318 Free PMC article.
-
Single-Molecule Light-Sheet Imaging of Suspended T Cells.Biophys J. 2018 May 8;114(9):2200-2211. doi: 10.1016/j.bpj.2018.02.044. Biophys J. 2018. PMID: 29742413 Free PMC article.
-
Machine learning predictions of T cell antigen specificity from intracellular calcium dynamics.Sci Adv. 2024 Mar 8;10(10):eadk2298. doi: 10.1126/sciadv.adk2298. Epub 2024 Mar 6. Sci Adv. 2024. PMID: 38446885 Free PMC article.
References
-
- Omilusik K. et al.. The Ca(v)1.4 calcium channel is a critical regulator of T cell receptor signaling and naive T cell homeostasis. Immunity 35, 349–360 (2011). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

