Changes in the Properties and Organization of Human Lens Lipid Membranes Occurring with Age
- PMID: 27791387
- PMCID: PMC5409882
- DOI: 10.1080/02713683.2016.1231325
Changes in the Properties and Organization of Human Lens Lipid Membranes Occurring with Age
Abstract
Purpose: This research was undertaken to document the changes in the organization and properties of human lens lipid membranes that occur with age.
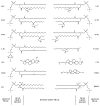
Methods: Human lens lipid membranes prepared from the total lipids extracted from clear lens cortices and nuclei of donors from age groups 0-20 and 21-40 years were investigated. An electron paramagnetic resonance technique and nitroxide spin labels (analogues of phospholipids and cholesterol) were used.
Results: Two distinct lipid domains, the phospholipid-cholesterol domain (PCD) and the pure cholesterol bilayer domain (CBD), were detected in all investigated membranes. Profiles of the acyl chain order, fluidity, hydrophobicity, and oxygen transport parameter across discriminated coexisting lipid domains were assessed. Independent of the age-related changes in phospholipid composition, the physical properties of the PCD remained the same for all age groups and were practically identical for cortical and nuclear membranes. However, the properties of pure CBDs changed significantly with the age of the donor and were related to the size of the CBD, which increased with the age of the donor and was greater in nuclear than in cortical membranes. A more detailed analysis revealed that the size of the CBD was determined mainly by the cholesterol content in the membrane.
Conclusions: This paper presents data from four age groups: 0-20, 21-40, 41-60, and 61-70 years. Data from age groups 41-60 and 61-70 years were published previously. Combining the previously published data with those data obtained in the present work allowed us to show the changes in the organization of cortical and nuclear lens lipid membranes as functions of age and cholesterol. It seems that the balance between age-related changes in membrane phospholipid composition and cholesterol content plays an integral role in the regulation of cholesterol-dependent processes in fiber cell membranes and in the maintenance of fiber cell membrane homeostasis.
Keywords: Cholesterol; cholesterol bilayer domain; cholesterol crystals; membrane domains; spin labeling.
Conflict of interest statement
The authors report no conflicts of interest.
Figures
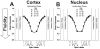

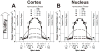
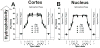
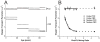
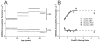

Similar articles
-
Lipid domains in intact fiber-cell plasma membranes isolated from cortical and nuclear regions of human eye lenses of donors from different age groups.Exp Eye Res. 2015 Mar;132:78-90. doi: 10.1016/j.exer.2015.01.018. Epub 2015 Jan 21. Exp Eye Res. 2015. PMID: 25617680 Free PMC article.
-
Cholesterol Bilayer Domains in the Eye Lens Health: A Review.Cell Biochem Biophys. 2017 Dec;75(3-4):387-398. doi: 10.1007/s12013-017-0812-7. Epub 2017 Jun 29. Cell Biochem Biophys. 2017. PMID: 28660427 Free PMC article. Review.
-
Properties of fiber cell plasma membranes isolated from the cortex and nucleus of the porcine eye lens.Exp Eye Res. 2012 Apr;97(1):117-29. doi: 10.1016/j.exer.2012.01.012. Epub 2012 Feb 2. Exp Eye Res. 2012. PMID: 22326289 Free PMC article.
-
Lipid-protein interactions in plasma membranes of fiber cells isolated from the human eye lens.Exp Eye Res. 2014 Mar;120:138-51. doi: 10.1016/j.exer.2014.01.018. Epub 2014 Jan 31. Exp Eye Res. 2014. PMID: 24486794 Free PMC article.
-
Functions of cholesterol and the cholesterol bilayer domain specific to the fiber-cell plasma membrane of the eye lens.J Membr Biol. 2012 Jan;245(1):51-68. doi: 10.1007/s00232-011-9412-4. Epub 2011 Dec 30. J Membr Biol. 2012. PMID: 22207480 Free PMC article. Review.
Cited by
-
Differences in the properties of porcine cortical and nuclear fiber cell plasma membranes revealed by saturation recovery EPR spin labeling measurements.Exp Eye Res. 2021 May;206:108536. doi: 10.1016/j.exer.2021.108536. Epub 2021 Mar 12. Exp Eye Res. 2021. PMID: 33716012 Free PMC article.
-
Tissue, cellular, and molecular level determinants for eye lens stiffness and elasticity.Front Ophthalmol (Lausanne). 2024 Aug 8;4:1456474. doi: 10.3389/fopht.2024.1456474. eCollection 2024. Front Ophthalmol (Lausanne). 2024. PMID: 39176256 Free PMC article. Review.
-
Factors Determining the Oxygen Permeability of Biological Membranes: Oxygen Transport Across Eye Lens Fiber-Cell Plasma Membranes.Adv Exp Med Biol. 2017;977:27-34. doi: 10.1007/978-3-319-55231-6_5. Adv Exp Med Biol. 2017. PMID: 28685424 Free PMC article. Review.
-
Mechanical properties of the high cholesterol-containing membrane: An AFM study.Biochim Biophys Acta Biomembr. 2021 Aug 1;1863(8):183625. doi: 10.1016/j.bbamem.2021.183625. Epub 2021 Apr 20. Biochim Biophys Acta Biomembr. 2021. PMID: 33891910 Free PMC article.
-
Cholesterol Content Regulates the Interaction of αA-, αB-, and α-Crystallin with the Model of Human Lens-Lipid Membranes.Int J Mol Sci. 2024 Feb 5;25(3):1923. doi: 10.3390/ijms25031923. Int J Mol Sci. 2024. PMID: 38339200 Free PMC article.
References
-
- Rafferty NS. Lens morphology. In: Maisel H, editor. The ocular lens:structure, function and pathology. New York: Marcel Dekker; 1985. pp. 1–60.
-
- Deeley JM, Mitchell TW, Wei X, Korth J, Nealon JR, Blanksby SJ, et al. Human lens lipids differ markedly from those of commonly used experimental animals. Biochim Biophys Acta. 2008;1781:288–298. - PubMed
-
- Huang L, Grami V, Marrero Y, Tang D, Yappert MC, Rasi V, et al. Human lens phospholipid changes with age and cataract. Invest Ophthalmol Vis Sci. 2005;46:1682–1689. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
