X-ray Diffraction Evidence for Low Force Actin-Attached and Rigor-Like Cross-Bridges in the Contractile Cycle
- PMID: 27792170
- PMCID: PMC5192421
- DOI: 10.3390/biology5040041
X-ray Diffraction Evidence for Low Force Actin-Attached and Rigor-Like Cross-Bridges in the Contractile Cycle
Abstract
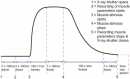
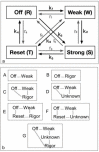
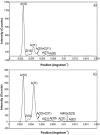
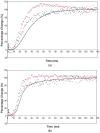
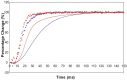
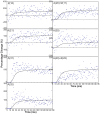
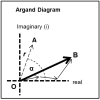
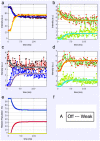
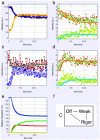
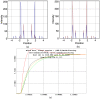
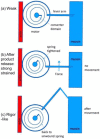
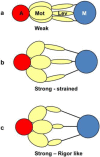
Defining the structural changes involved in the myosin cross-bridge cycle on actin in active muscle by X-ray diffraction will involve recording of the whole two dimensional (2D) X-ray diffraction pattern from active muscle in a time-resolved manner. Bony fish muscle is the most highly ordered vertebrate striated muscle to study. With partial sarcomere length (SL) control we show that changes in the fish muscle equatorial A-band (10) and (11) reflections, along with (10)/(11) intensity ratio and the tension, are much more rapid than without such control. Times to 50% change with SL control were 19.5 (±2.0) ms, 17.0 (±1.1) ms, 13.9 (±0.4) ms and 22.5 (±0.8) ms, respectively, compared to 25.0 (±3.4) ms, 20.5 (±2.6) ms, 15.4 (±0.6) ms and 33.8 (±0.6) ms without control. The (11) intensity and the (10)/(11) intensity ratio both still change ahead of tension, supporting the likelihood of the presence of a head population close to or on actin, but producing little or no force, in the early stages of the contractile cycle. Higher order equatorials (e.g., (30), (31), and (32)), more sensitive to crossbridge conformation and distribution, also change very rapidly and overshoot their tension plateau values by a factor of around two, well before the tension plateau has been reached, once again indicating an early low-force cross-bridge state in the contractile cycle. Modelling of these intensity changes suggests the presence of probably two different actin-attached myosin head structural states (mainly low-force attached and rigor-like). No more than two main attached structural states are necessary and sufficient to explain the observations. We find that 48% of the heads are off actin giving a resting diffraction pattern, 20% of heads are in the weak binding conformation and 32% of the heads are in the strong (rigor-like) state. The strong states account for 96% of the tension at the tetanus plateau.
Keywords: crossbridge time-course modelling; equatorial X-ray diffraction; myosin cross-bridge cycle; sarcomere length control; tetanus rising phase.
Conflict of interest statement
The authors declare no conflict of interest. The founding sponsors had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, and in the decision to publish the results.
Figures
Similar articles
-
Myosin Cross-Bridge Behaviour in Contracting Muscle-The T1 Curve of Huxley and Simmons (1971) Revisited.Int J Mol Sci. 2019 Oct 2;20(19):4892. doi: 10.3390/ijms20194892. Int J Mol Sci. 2019. PMID: 31581677 Free PMC article.
-
Crossbridge states in isometrically contracting fish muscle: evidence for swinging of myosin heads on actin.Adv Biophys. 1991;27:45-61. doi: 10.1016/0065-227x(91)90007-z. Adv Biophys. 1991. PMID: 1755367
-
Evidence for structurally different attached states of myosin cross-bridges on actin during contraction of fish muscle.Biophys J. 1992 Aug;63(2):387-96. doi: 10.1016/S0006-3495(92)81613-2. Biophys J. 1992. PMID: 1420885 Free PMC article.
-
Monitoring the myosin crossbridge cycle in contracting muscle: steps towards 'Muscle-the Movie'.J Muscle Res Cell Motil. 2019 Jun;40(2):77-91. doi: 10.1007/s10974-019-09543-9. Epub 2019 Jul 20. J Muscle Res Cell Motil. 2019. PMID: 31327096 Free PMC article. Review.
-
Time-resolved studies of crossbridge movement: why use X-rays? Why use fish muscle?Adv Exp Med Biol. 1993;332:435-48; discussion 448-50. doi: 10.1007/978-1-4615-2872-2_40. Adv Exp Med Biol. 1993. PMID: 8109356 Review.
Cited by
-
Geometric frustration in the myosin superlattice of vertebrate muscle.J R Soc Interface. 2021 Dec;18(185):20210585. doi: 10.1098/rsif.2021.0585. Epub 2021 Dec 15. J R Soc Interface. 2021. PMID: 34905966 Free PMC article.
-
Myosin Cross-Bridge Behaviour in Contracting Muscle-The T1 Curve of Huxley and Simmons (1971) Revisited.Int J Mol Sci. 2019 Oct 2;20(19):4892. doi: 10.3390/ijms20194892. Int J Mol Sci. 2019. PMID: 31581677 Free PMC article.
-
Different Myosin Head Conformations in Bony Fish Muscles Put into Rigor at Different Sarcomere Lengths.Int J Mol Sci. 2018 Jul 18;19(7):2091. doi: 10.3390/ijms19072091. Int J Mol Sci. 2018. PMID: 30022010 Free PMC article.
-
Fast myosin binding protein C knockout in skeletal muscle alters length-dependent activation and myofilament structure.Commun Biol. 2024 May 27;7(1):648. doi: 10.1038/s42003-024-06265-8. Commun Biol. 2024. PMID: 38802450 Free PMC article.
-
Using Multiscale Simulations as a Tool to Interpret Equatorial X-ray Fiber Diffraction Patterns from Skeletal Muscle.Int J Mol Sci. 2023 May 9;24(10):8474. doi: 10.3390/ijms24108474. Int J Mol Sci. 2023. PMID: 37239821 Free PMC article.
References
-
- Squire J.M., Al-Khayat H.A., Knupp C., Luther P.K. Molecular architecture in muscle contractile assemblies. Adv. Protein Chem. 2005;71:17–87. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

