Endoplasmic reticulum stress-induced neuronal inflammatory response and apoptosis likely plays a key role in the development of diabetic encephalopathy
- PMID: 27793043
- PMCID: PMC5346653
- DOI: 10.18632/oncotarget.12925
Endoplasmic reticulum stress-induced neuronal inflammatory response and apoptosis likely plays a key role in the development of diabetic encephalopathy
Abstract
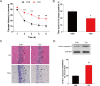
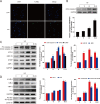
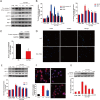
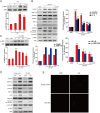
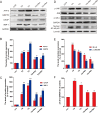
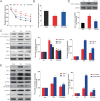
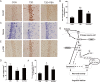
We assumed that diabetic encephalopathy (DEP) may be induced by endoplasmic reticulum (ER)-mediated inflammation and apoptosis in central nervous system. To test this notion, here we investigated the neuronal ER stress and associated inflammation and apoptosis in a type 2 diabetes model induced with high-fat diet/streptozotocin in Sprague-Dawley rats. Elevated expressions of ER stress markers, including glucose-regulated protein 78 (GRP78), activating transcription factor-6 (ATF-6), X-box binding protein-1 (XBP-1), and C/EBP homologous protein, and phosphor-Jun N-terminal kinase (p-JNK) were evident in the hippocampus CA1 of diabetic rats. These changes were also accompanied with the activation of NF-κB and the increased levels of inflammatory cytokines, tumor necrosis factor-α (TNF-α) and Interleukin-6 (IL-6). Mechanistic study with in vitro cultured hippocampus neurons exposed to high glucose (HG), which induced a diabetes-like effects, shown by increased ER stress, JNK and NF-κB activation, and inflammatory response. Inhibition of ER stress by 4-phenylbutyrate (4-PBA) or blockade of JNK activity by specific inhibitor or transfection of DN-JNK attenuated HG-induced inflammation and associated apoptosis. To validate the in vitro finding, in vivo application of 4-PBA resulted in a significant reduction of diabetes-induced neuronal ER stress, inflammation and cell death, leading to the prevention of DEP. These results suggest that diabetes-induced neuronal ER stress plays the critical role for diabetes-induced neuronal inflammation and cell death, leading to the development of DEP.
Keywords: ER stress; apoptosis; diabetes; inflammation; p-JNK.
Conflict of interest statement
The authors do not have any conflict interest to disclosure
Figures

References
-
- Sima AA, Li ZG. The effect of C-peptide on cognitive dysfunction and hippocampal apoptosis in type 1 diabetic rats. Diabetes. 2005;54:1497–1505. - PubMed
-
- Mijnhout GS, Scheltens P, Diamant M, Biessels GJ, Wessels AM, Simsek S, Snoek FJ, Heine RJ. Diabetic encephalopathy: A concept in need of a definition. Diabetologia. 2006;49:1447–1448. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

