Biodistribution of Self-Assembling Polymer-Gemcitabine Conjugate after Systemic Administration into Orthotopic Pancreatic Tumor Bearing Mice
- PMID: 27798825
- PMCID: PMC5997252
- DOI: 10.1021/acs.molpharmaceut.6b00929
Biodistribution of Self-Assembling Polymer-Gemcitabine Conjugate after Systemic Administration into Orthotopic Pancreatic Tumor Bearing Mice
Abstract
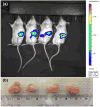
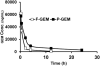
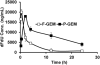
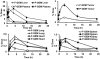
Therapeutic efficacy of gemcitabine (GEM) is severely limited due to its rapid metabolism by enzymatic deamination in vivo. We recently determined its therapeutic efficacy before (F-GEM) and after conjugation to poly(ethylene glycol)-block-poly(2-methyl-2-carboxyl-propylene carbonate) (mPEG-b-PCC-g-GEM-g-DC, abbreviated as P-GEM) in subcutaneous and orthotopic pancreatic tumor bearing mice. In this study, pharmacokinetic (PK) parameters and biodistribution profiles of F-GEM and P-GEM were determined after intravenous injection into orthotopic pancreatic tumor bearing NSG mice. To assess the short-term toxicity, the levels of hematological, hepatic, and renal injury markers were measured after 24 h postadministration into these mice. P-GEM was distributed to all the major organs, with higher accumulation in the liver, spleen, and tumor compared to F-GEM. Area under the curve (AUC), elimination half-life (t1/2), and mean residence time (MRT) of P-GEM treated group were significantly higher compared to those of F-GEM treated group: 246,425 ± 1605 vs 83,591 ± 1844 ng/mL × h as AUC, 5.77 ± 2.02 vs 1.99 ± 0.09 h as t1/2, and 4.45 ± 0.15 vs 1.12 ± 0.13 h as MRT. Further, P-GEM exhibited negligible systemic toxicity as evidenced by almost similar alanine aminotransferase (ALT) and aspartate aminotransferase (AST) values for both P-GEM and F-GEM. These results suggest that P-GEM protects GEM from degradation and provides sustained drug release, resulting in enhanced GEM delivery to the tumor by more than 2.5-fold compared to F-GEM. Hence, P-GEM is a promising gemcitabine conjugated polymeric micelle for treating pancreatic cancer.
Keywords: biodistribution; gemcitabine; pancreatic cancer; pharmacokinetics; polymeric micelles.
Figures
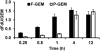
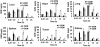
Similar articles
-
EGFR-Targeted Cationic Polymeric Mixed Micelles for Codelivery of Gemcitabine and miR-205 for Treating Advanced Pancreatic Cancer.Mol Pharm. 2017 Sep 5;14(9):3121-3133. doi: 10.1021/acs.molpharmaceut.7b00355. Epub 2017 Jul 31. Mol Pharm. 2017. PMID: 28719220 Free PMC article.
-
EGFR-Targeted Polymeric Mixed Micelles Carrying Gemcitabine for Treating Pancreatic Cancer.Biomacromolecules. 2016 Jan 11;17(1):301-13. doi: 10.1021/acs.biomac.5b01419. Epub 2015 Dec 17. Biomacromolecules. 2016. PMID: 26626700 Free PMC article.
-
Micelle Mixtures for Coadministration of Gemcitabine and GDC-0449 To Treat Pancreatic Cancer.Mol Pharm. 2016 Jun 6;13(6):1822-32. doi: 10.1021/acs.molpharmaceut.5b00971. Epub 2016 Apr 27. Mol Pharm. 2016. PMID: 26981724 Free PMC article.
-
Chemosensitization and inhibition of pancreatic cancer stem cell proliferation by overexpression of microRNA-205.Cancer Lett. 2017 Aug 28;402:1-8. doi: 10.1016/j.canlet.2017.05.007. Epub 2017 May 20. Cancer Lett. 2017. PMID: 28536008 Free PMC article.
-
Nanotechnology for delivery of gemcitabine to treat pancreatic cancer.Biomed Pharmacother. 2017 Apr;88:635-643. doi: 10.1016/j.biopha.2017.01.071. Epub 2017 Jan 28. Biomed Pharmacother. 2017. PMID: 28142120 Review.
Cited by
-
Human bone marrow mesenchymal stem cell-derived exosomes loaded with gemcitabine inhibit pancreatic cancer cell proliferation by enhancing apoptosis.World J Gastrointest Oncol. 2024 Sep 15;16(9):4006-4013. doi: 10.4251/wjgo.v16.i9.4006. World J Gastrointest Oncol. 2024. PMID: 39350998 Free PMC article.
-
Gemcitabine-retinoid prodrug loaded nanoparticles display in vitro antitumor efficacy towards drug-resilient human PANC-1 pancreatic cancer cells.Mater Sci Eng C Mater Biol Appl. 2020 Dec;117:111251. doi: 10.1016/j.msec.2020.111251. Epub 2020 Jul 4. Mater Sci Eng C Mater Biol Appl. 2020. PMID: 32919625 Free PMC article.
-
EGFR-Targeted Cationic Polymeric Mixed Micelles for Codelivery of Gemcitabine and miR-205 for Treating Advanced Pancreatic Cancer.Mol Pharm. 2017 Sep 5;14(9):3121-3133. doi: 10.1021/acs.molpharmaceut.7b00355. Epub 2017 Jul 31. Mol Pharm. 2017. PMID: 28719220 Free PMC article.
-
CD34+ progenitor-derived NK cell and gemcitabine combination therapy increases killing of ovarian cancer cells in NOD/SCID/IL2Rgnull mice.Oncoimmunology. 2021 Oct 1;10(1):1981049. doi: 10.1080/2162402X.2021.1981049. eCollection 2021. Oncoimmunology. 2021. PMID: 34616589 Free PMC article.
-
Pharmacokinetics and biodistribution of polymeric micelles containing miRNA and small-molecule drug in orthotopic pancreatic tumor-bearing mice.Theranostics. 2018 Jul 5;8(15):4033-4049. doi: 10.7150/thno.24945. eCollection 2018. Theranostics. 2018. PMID: 30128034 Free PMC article.
References
-
- Berger AC, Meszoely IM, Ross EA, Watson JC, Hoffman JP. Undetectable preoperative levels of serum CA 19–9 correlate with improved survival for patients with resectable pancreatic adenocarcinoma. Ann Surg Oncol. 2004;11:644–649. - PubMed
-
- Shore S, Vimalachandran D, Raraty MG, Ghaneh P. Cancer in the elderly: pancreatic cancer. Surg Oncol. 2004;13:201–210. - PubMed
-
- Wang Z, Li Y, Ahmad A, Banerjee S, Azmi AS, Kong D, Sarkar FH. Pancreatic cancer: understanding and overcoming chemoresistance. Nat Rev Gastroenterol Hepatol. 2011;8:27–33. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

