Fatty alcohol production in Lipomyces starkeyi and Yarrowia lipolytica
- PMID: 27800013
- PMCID: PMC5078963
- DOI: 10.1186/s13068-016-0647-2
Fatty alcohol production in Lipomyces starkeyi and Yarrowia lipolytica
Abstract
Background: Current biological pathways to produce biofuel intermediates amenable to separations and catalytic upgrading to hydrocarbon fuels are not cost effective. Previously, oleaginous yeasts have been investigated primarily for lipid production. However, yeasts store neutral lipids intracellularly making recovery difficult and expensive. In addition, once recovered from the cells, lipids are difficult to blend directly with the existing fuels without upgrading. We have, therefore, begun to investigate secreted fatty acid-derived products which can be easily recovered and upgraded to fuels.
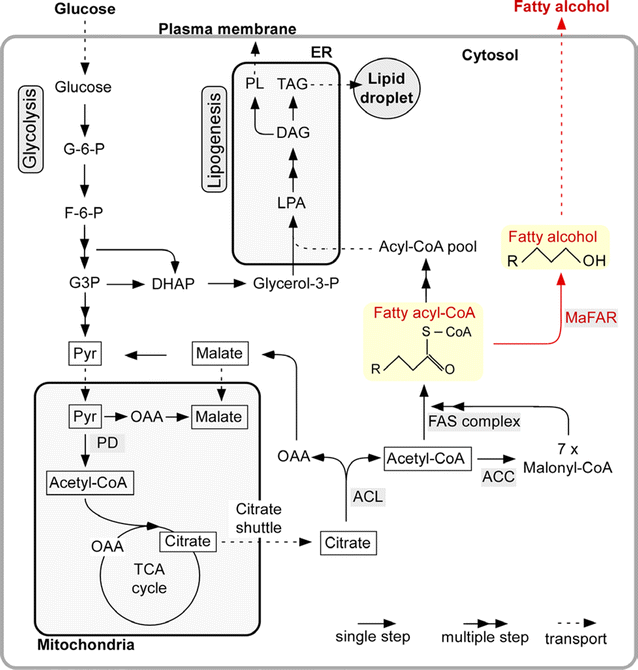
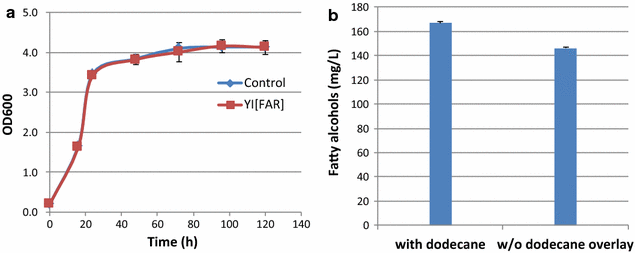
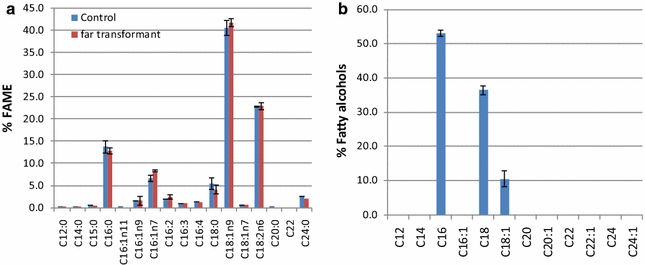
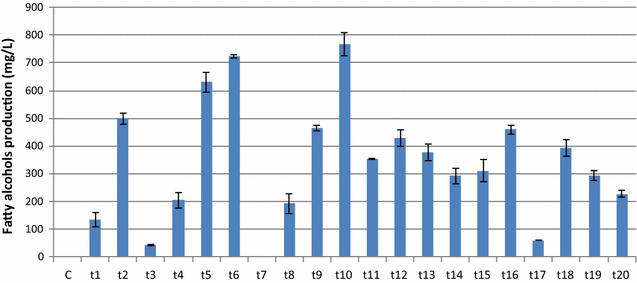
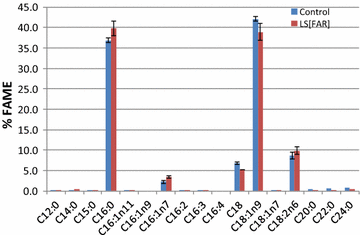
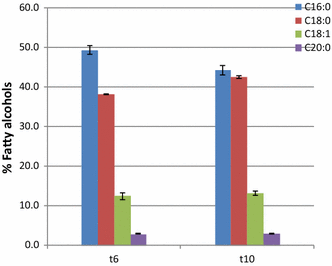
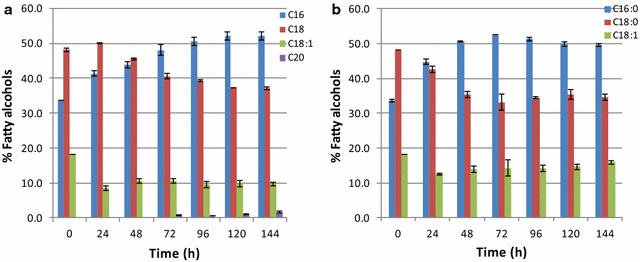
Results: In this study, we successfully demonstrate the production of fatty alcohols by the oleaginous yeasts, Yarrowia lipolytica and Lipomyces starkeyi, through expression of the fatty acyl-CoA reductase gene from Marinobactor aquaeolei VT8. This strategy resulted in the production of 167 and 770 mg/L of fatty alcohols in shake flask from Y. lipolytica and L starkeyi, respectively. When using a dodecane overlay during fermentation, 92 and 99% of total fatty alcohols produced by Y. lipolytica and L. starkeyi, respectively, were extracted into the dodecane phase, which compares favorably to the 3 and 50% recovered, respectively, without the dodecane layer. In both oleaginous yeasts, long chain length, saturated fatty alcohols, i.e., hexadecanol (C16:0) and octadecanol (C18:0), were predominant and accounted for more than 85% of the total fatty alcohols produced. To the best of our knowledge, this is the first report of fatty alcohol production in L. starkeyi.
Conclusion: This work demonstrates that the oleaginous yeasts, Y. lipolytica and L. starkeyi, can serve as platform organisms for the production of fatty acid-derived biofuels and bioproducts.
Keywords: Fatty alcohols; Lipomyces starkeyi; Metabolic engineering; Oleaginous yeasts; Yarrowia lipolytica.
Figures

Similar articles
-
Lipid production by yeasts growing on biodiesel-derived crude glycerol: strain selection and impact of substrate concentration on the fermentation efficiency.J Appl Microbiol. 2015 Apr;118(4):911-27. doi: 10.1111/jam.12736. Epub 2015 Feb 12. J Appl Microbiol. 2015. PMID: 25626733
-
Expression and secretion of fungal endoglucanase II and chimeric cellobiohydrolase I in the oleaginous yeast Lipomyces starkeyi.Microb Cell Fact. 2017 Jul 24;16(1):126. doi: 10.1186/s12934-017-0742-5. Microb Cell Fact. 2017. PMID: 28738851 Free PMC article.
-
Optimization of C16 and C18 fatty alcohol production by an engineered strain of Lipomyces starkeyi.J Ind Microbiol Biotechnol. 2018 Jan;45(1):1-14. doi: 10.1007/s10295-017-1985-1. Epub 2017 Oct 26. J Ind Microbiol Biotechnol. 2018. PMID: 29076046
-
Oleaginous yeast for biofuel and oleochemical production.Curr Opin Biotechnol. 2019 Jun;57:73-81. doi: 10.1016/j.copbio.2019.02.011. Epub 2019 Mar 12. Curr Opin Biotechnol. 2019. PMID: 30875587 Review.
-
Carotenoid Production in Oleaginous Yeasts.Adv Exp Med Biol. 2021;1261:153-163. doi: 10.1007/978-981-15-7360-6_12. Adv Exp Med Biol. 2021. PMID: 33783737 Review.
Cited by
-
Exploiting nonionic surfactants to enhance fatty alcohol production in Rhodosporidium toruloides.Biotechnol Bioeng. 2020 May;117(5):1418-1425. doi: 10.1002/bit.27285. Epub 2020 Feb 11. Biotechnol Bioeng. 2020. PMID: 31981215 Free PMC article.
-
Non-homologous End Joining-Mediated Insertional Mutagenesis Reveals a Novel Target for Enhancing Fatty Alcohols Production in Yarrowia lipolytica.Front Microbiol. 2022 Apr 25;13:898884. doi: 10.3389/fmicb.2022.898884. eCollection 2022. Front Microbiol. 2022. PMID: 35547152 Free PMC article.
-
System analysis of Lipomyces starkeyi during growth on various plant-based sugars.Appl Microbiol Biotechnol. 2022 Sep;106(17):5629-5642. doi: 10.1007/s00253-022-12084-w. Epub 2022 Jul 30. Appl Microbiol Biotechnol. 2022. PMID: 35906440
-
Production of Long Chain Fatty Alcohols Found in Bumblebee Pheromones by Yarrowia lipolytica.Front Bioeng Biotechnol. 2021 Jan 8;8:593419. doi: 10.3389/fbioe.2020.593419. eCollection 2020. Front Bioeng Biotechnol. 2021. PMID: 33490049 Free PMC article.
-
What are the 100 most cited fungal genera?Stud Mycol. 2024 Jul;108:1-411. doi: 10.3114/sim.2024.108.01. Epub 2024 Jul 15. Stud Mycol. 2024. PMID: 39100921 Free PMC article.
References
-
- Wolf DA. Nonconventional yeasts in biotechnology, a handbook. Recherche. 1996;67:02.
LinkOut - more resources
Full Text Sources
Other Literature Sources

