De novo assembly of plant body plan: a step ahead of Deadpool
- PMID: 27800169
- PMCID: PMC5084358
- DOI: 10.1002/reg2.68
De novo assembly of plant body plan: a step ahead of Deadpool
Abstract
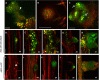
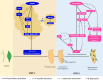
While in the movie Deadpool it is possible for a human to recreate an arm from scratch, in reality plants can even surpass that. Not only can they regenerate lost parts, but also the whole plant body can be reborn from a few existing cells. Despite the decades old realization that plant cells possess the ability to regenerate a complete shoot and root system, it is only now that the underlying mechanisms are being unraveled. De novo plant regeneration involves the initiation of regenerative mass, acquisition of the pluripotent state, reconstitution of stem cells and assembly of regulatory interactions. Recent studies have furthered our understanding on the making of a complete plant system in the absence of embryonic positional cues. We review the recent studies probing the molecular mechanisms of de novo plant regeneration in response to external inductive cues and our current knowledge of direct reprogramming of root to shoot and vice versa. We further discuss how de novo regeneration can be exploited to meet the demands of green culture industries and to serve as a general model to address the fundamental questions of regeneration across the plant kingdom.
Keywords: auxin; callus; cytokinin; de novo organogenesis; pluripotency; regeneration; stem cells; transdifferentiation.
Figures



Similar articles
-
Ectopic expression of WOX5 promotes cytokinin signaling and de novo shoot regeneration.Plant Cell Rep. 2022 Dec;41(12):2415-2422. doi: 10.1007/s00299-022-02932-4. Epub 2022 Oct 11. Plant Cell Rep. 2022. PMID: 36219248
-
Plant stem cells and de novo organogenesis.New Phytol. 2018 Jun;218(4):1334-1339. doi: 10.1111/nph.15106. Epub 2018 Mar 25. New Phytol. 2018. PMID: 29574802 Review.
-
De novo shoot organogenesis during plant regeneration.J Exp Bot. 2020 Jan 1;71(1):63-72. doi: 10.1093/jxb/erz395. J Exp Bot. 2020. PMID: 31504722 Review.
-
Competence and regulatory interactions during regeneration in plants.Front Plant Sci. 2014 Apr 11;5:142. doi: 10.3389/fpls.2014.00142. eCollection 2014. Front Plant Sci. 2014. PMID: 24782880 Free PMC article. Review.
-
Control of Cell Fate Reprogramming Towards De Novo Shoot Organogenesis.Plant Cell Physiol. 2018 Apr 1;59(4):708-714. doi: 10.1093/pcp/pcx207. Plant Cell Physiol. 2018. PMID: 29294130 Review.
Cited by
-
What Makes Adventitious Roots?Plants (Basel). 2019 Jul 22;8(7):240. doi: 10.3390/plants8070240. Plants (Basel). 2019. PMID: 31336687 Free PMC article. Review.
-
Pivotal role of LBD16 in root and root-like organ initiation.Cell Mol Life Sci. 2018 Sep;75(18):3329-3338. doi: 10.1007/s00018-018-2861-5. Epub 2018 Jun 25. Cell Mol Life Sci. 2018. PMID: 29943076 Free PMC article. Review.
-
Natural Variation in Plant Pluripotency and Regeneration.Plants (Basel). 2020 Sep 24;9(10):1261. doi: 10.3390/plants9101261. Plants (Basel). 2020. PMID: 32987766 Free PMC article. Review.
-
Identification of Shoot Differentiation-Related Genes in Populus euphratica Oliv.Genes (Basel). 2019 Dec 11;10(12):1034. doi: 10.3390/genes10121034. Genes (Basel). 2019. PMID: 31835855 Free PMC article.
-
Integrating the Roles for Cytokinin and Auxin in De Novo Shoot Organogenesis: From Hormone Uptake to Signaling Outputs.Int J Mol Sci. 2021 Aug 9;22(16):8554. doi: 10.3390/ijms22168554. Int J Mol Sci. 2021. PMID: 34445260 Free PMC article. Review.
References
-
- Ahkami, A. H. , Lischewski, S. , Haensch, K. T. , Porfirova, S. , Hofmann, J. , Rolletschek, H. , …, & Hajirezaei, M. R . (2009). Molecular physiology of adventitious root formation in Petunia hybrida cuttings: involvement of wound response and primary metabolism. New Phytologist, 181(3), 613–625. - PubMed
-
- Aida, M. , Beis, D. , Heidstra, R. , Willemsen, V. , Blilou, I. , Galinha, C. , …, & Scheres, B. (2004). The PLETHORA genes mediate patterning of the Arabidopsis root stem cell niche. Cell, 119(1), 109–120. doi:10.1016/j.cell.2004.09.018 - DOI - PubMed
-
- Akama, K. , Shiraishi, H. , Ohta, S. , Nakamura, K. , Okada, K. , & Shimura, Y. (1992). Efficient transformation of Arabidopsis thaliana: comparison of the efficiencies with various organs, plant ecotypes and Agrobacterium strains. Plant Cell Reports, 12, 7–11. doi:10.1007/BF00232413 - DOI - PubMed
-
- Anzola, J. M. , Sieberer, T. , Ortbauer, M. , Butt, H. , Korbei, B. , Weinhofer, I. , …, & Luschnig, C. (2010). Putative Arabidopsis transcriptional adaptor protein (PROPORZ1) is required to modulate histone acetylation in response to auxin. Proceedings of the National Academy of Sciences USA, 107(22), 10308–10313. doi:10.1073/pnas.0913918107 - DOI - PMC - PubMed
-
- Atta, R. , Laurens, L. , Boucheron‐Dubuisson, E. , Guivarc'h, A. , Carnero, E. , Giraudat‐Pautot, V. , …, & Chriqui, D. (2009). Pluripotency of Arabidopsis xylem pericycle underlies shoot regeneration from root and hypocotyl explants grown in vitro. The Plant Journal, 57(4), 626–644. doi:10.1111/j.1365-313X.2008.03715.x - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
