Clinical and imaging characteristics of late onset mitochondrial membrane protein-associated neurodegeneration (MPAN)
- PMID: 27801611
- PMCID: PMC5568540
- DOI: 10.1080/13554794.2016.1247458
Clinical and imaging characteristics of late onset mitochondrial membrane protein-associated neurodegeneration (MPAN)
Abstract
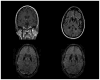
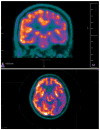
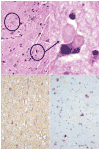
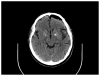
Young onset dementias present significant diagnostic challenges. We present the case of a 35-year-old Kuwaiti man with social withdrawal, drowsiness, irritability, anxiety, aphasia, memory loss, hypereflexia, and Parkinsonism. Brain MRI showed bilateral symmetric gradient echo hypointensities in the globi pallidi and substantiae nigrae. Left cortical hypometabolism was seen on brain fluorodeoxyglucose positron emission tomography. A cortical brain biopsy revealed a high Lewy body burden. Genetic testing revealed a homozygous p.T11M mutation in the C19orf12 gene consistent with mitochondrial membrane protein-associated neurodegeneration. This is the oldest onset age of MPAN reported.
Keywords: Lewy body; Mitochondrial membrane protein-associatedneurodegeneration (MPAN); Parkinsonism; neurodegeneration with brain iron accumulation (NBIA); whole exome sequencing.
Conflict of interest statement
The authors have no financial or nonfinancial relationships to disclose.
Figures

Similar articles
-
The p.Thr11Met mutation in c19orf12 is frequent among adult Turkish patients with MPAN.Parkinsonism Relat Disord. 2017 Jun;39:64-70. doi: 10.1016/j.parkreldis.2017.03.012. Epub 2017 Mar 21. Parkinsonism Relat Disord. 2017. PMID: 28347615
-
Evolution and novel radiological changes of neurodegeneration associated with mutations in C19orf12.Parkinsonism Relat Disord. 2017 Jun;39:71-76. doi: 10.1016/j.parkreldis.2017.03.013. Epub 2017 Mar 21. Parkinsonism Relat Disord. 2017. PMID: 28347614
-
[Pedigree analysis of C19ORF12 p.Asp18Tyr mutation in a family with mitochondrial membrane protein associated neurodegeneration].Zhonghua Yi Xue Za Zhi. 2019 Oct 8;99(37):2926-2931. doi: 10.3760/cma.j.issn.0376-2491.2019.37.011. Zhonghua Yi Xue Za Zhi. 2019. PMID: 31607023 Chinese.
-
Mitochondrial membrane protein-associated neurodegeneration (MPAN).Int Rev Neurobiol. 2013;110:73-84. doi: 10.1016/B978-0-12-410502-7.00004-1. Int Rev Neurobiol. 2013. PMID: 24209434 Review.
-
NBIA Syndromes: A Step Forward from the Previous Knowledge.Neurol India. 2021 Sep-Oct;69(5):1380-1388. doi: 10.4103/0028-3886.329603. Neurol India. 2021. PMID: 34747818 Review.
Cited by
-
Emerging Disease-Modifying Therapies in Neurodegeneration With Brain Iron Accumulation (NBIA) Disorders.Front Neurol. 2021 Apr 15;12:629414. doi: 10.3389/fneur.2021.629414. eCollection 2021. Front Neurol. 2021. PMID: 33935938 Free PMC article. Review.
-
Mitochondrial Dysfunction, Oxidative Stress and Neuroinflammation in Neurodegeneration with Brain Iron Accumulation (NBIA).Antioxidants (Basel). 2020 Oct 20;9(10):1020. doi: 10.3390/antiox9101020. Antioxidants (Basel). 2020. PMID: 33092153 Free PMC article. Review.
-
C19orf12 ablation causes ferroptosis in mitochondrial membrane protein-associated with neurodegeneration.Free Radic Biol Med. 2022 Mar;182:23-33. doi: 10.1016/j.freeradbiomed.2022.02.006. Epub 2022 Feb 17. Free Radic Biol Med. 2022. PMID: 35182730 Free PMC article.
-
A novel C19ORF12 mutation in two MPAN sisters treated with deferiprone.BMC Neurol. 2023 Mar 31;23(1):134. doi: 10.1186/s12883-023-03172-z. BMC Neurol. 2023. PMID: 37004026 Free PMC article.
-
An Updated Overview of the Magnetic Resonance Imaging of Brain Iron in Movement Disorders.Behav Neurol. 2022 Feb 24;2022:3972173. doi: 10.1155/2022/3972173. eCollection 2022. Behav Neurol. 2022. PMID: 35251368 Free PMC article. Review.
References
-
- Al-Semari A, Bohlega S. Autosomal-recessive syndrome with alopecia, hypogonadism, progressive extra-pyramidal disorder, white matter disease, sensory neural deafness, diabetes mellitus, and low IGF1. American Journal of Medical Genetics Part A. 2007;143a:149–160. doi: 10.1002/ajmg.a.31497. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
