Transition Metal Intercalators as Anticancer Agents-Recent Advances
- PMID: 27809241
- PMCID: PMC5133819
- DOI: 10.3390/ijms17111818
Transition Metal Intercalators as Anticancer Agents-Recent Advances
Abstract
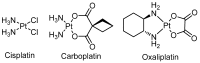
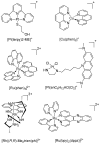
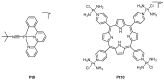
The diverse anticancer utility of cisplatin has stimulated significant interest in the development of additional platinum-based therapies, resulting in several analogues receiving clinical approval worldwide. However, due to structural and mechanistic similarities, the effectiveness of platinum-based therapies is countered by severe side-effects, narrow spectrum of activity and the development of resistance. Nonetheless, metal complexes offer unique characteristics and exceptional versatility, with the ability to alter their pharmacology through facile modifications of geometry and coordination number. This has prompted the search for metal-based complexes with distinctly different structural motifs and non-covalent modes of binding with a primary aim of circumventing current clinical limitations. This review discusses recent advances in platinum and other transition metal-based complexes with mechanisms of action involving intercalation. This mode of DNA binding is distinct from cisplatin and its derivatives. The metals focused on in this review include Pt, Ru and Cu along with examples of Au, Ni, Zn and Fe complexes; these complexes are capable of DNA intercalation and are highly biologically active.
Keywords: DNA; DNA binding; cancer; cytotoxicity; intercalate; platinum; transition metals.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

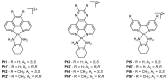


References
-
- Provencher-Mandeville J., Debnath C., Mandal S.K., Leblanc V., Parent S., Asselin É., Bérubé G. Design, synthesis and biological evaluation of estradiol-PEG-linked Platinum(II) hybrid molecules: Comparative molecular modeling study of three distinct families of hybrids. Steroids. 2011;76:94–103. doi: 10.1016/j.steroids.2010.09.004. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

