Acute neuroinflammation provokes intracellular acidification in mouse hippocampus
- PMID: 27809864
- PMCID: PMC5094044
- DOI: 10.1186/s12974-016-0747-8
Acute neuroinflammation provokes intracellular acidification in mouse hippocampus
Abstract
Background: Maintaining pH levels within the physiological norm is an important component of brain homeostasis. However, in some pathological or physiological conditions, the capacity of the pH regulatory system could be overpowered by various factors resulting in a transient or permanent alteration in pH levels. Such changes are often observed in pathological conditions associated with neuroinflammation. We hypothesized that neuroinflammation itself is a factor affecting pH levels in neural tissue. To assess this hypothesis, we examined the effects of acute LPS-induced neuroinflammation on intra- and extracellular pH (pHi and pHo) levels in the CA1 region of mouse hippocampus.
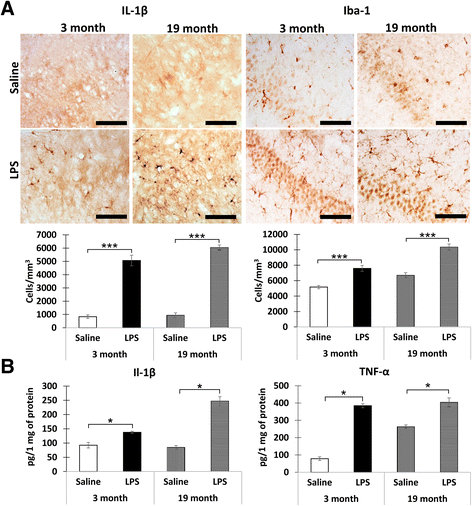
Methods: Acute neuroinflammation was induced using two approaches: (1) in vivo by i.p. injections of LPS (5 mg/kg) and (2) in vitro by incubating hippocampal slices of naïve animals in the LPS-containing media (1 μg/mL, 1 h at 35 °C). Standard techniques were used to prepare hippocampal slices. pHi was measured using ratiometric pH-sensitive fluorescent dye BCECF-AM. pHo was assessed using calibrated pH-sensitive micropipettes. The presence of neuroinflammation was verified with immunohistochemistry (IL-1β and Iba1) and ELISA (IL-1β and TNF-α).
Results: A significant reduction of pHi was observed in the slices of the LPS-injected 3-month-old (LPS 7.13 ± 0.03; Sal 7.22 ± 0.03; p = 0.043, r = 0.43) and 19-month-old (LPS 6.78 ± 0.08; Sal 7.13 ± 0.03; p = 0.0001, r = 0.32) mice. In contrast, the levels of pHo within the slice, measured in 19-month-old animals, were not affected (LPS 7.27 ± 0.02; Sal 7.26 ± 0.02; p = 0.6, r = 0.13). A reduction of pHi was also observed in the LPS-treated slices during the interval 3.5-7 h after the LPS exposure (LPS 6.92 ± 0.07; Veh 7.28 ± 0.05; p = 0.0001, r = 0.46).
Conclusions: Acute LPS-induced neuroinflammation results in a significant intracellular acidification of the CA1 neurons in mouse hippocampus, while the pHo remains largely unchanged. Such changes may represent a specific protective reaction of neural tissue in unfavorable external conditions or be a part of the pathological process.
Keywords: Acidification; BCECF; Extracellular pH; Hippocampus; IL-1β; Iba1; Intracellular pH; LPS; Mice; Neuroinflammation.
Figures




References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

