Mfge8 regulates enterocyte lipid storage by promoting enterocyte triglyceride hydrolase activity
- PMID: 27812539
- PMCID: PMC5085605
- DOI: 10.1172/jci.insight.87418
Mfge8 regulates enterocyte lipid storage by promoting enterocyte triglyceride hydrolase activity
Abstract
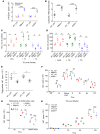
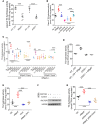
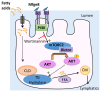
The small intestine has an underappreciated role as a lipid storage organ. Under conditions of high dietary fat intake, enterocytes can minimize the extent of postprandial lipemia by storing newly absorbed dietary fat in cytoplasmic lipid droplets. Lipid droplets can be subsequently mobilized for the production of chylomicrons. The mechanisms that regulate this process are poorly understood. We report here that the milk protein Mfge8 regulates hydrolysis of cytoplasmic lipid droplets in enterocytes after interacting with the αvβ3 and αvβ5 integrins. Mice deficient in Mfge8 or the αvβ3 and αvβ5 integrins accumulate excess cytoplasmic lipid droplets after a fat challenge. Mechanistically, interruption of the Mfge8-integrin axis leads to impaired enterocyte intracellular triglyceride hydrolase activity in vitro and in vivo. Furthermore, Mfge8 increases triglyceride hydrolase activity through a PI3 kinase/mTORC2-dependent signaling pathway. These data identify a key role for Mfge8 and the αvβ3 and αvβ5 integrins in regulating enterocyte lipid processing.
Conflict of interest statement
The authors have declared that no conflict of interest exists.
Figures


References
-
- Federico LM, Naples M, Taylor D, Adeli K. Intestinal insulin resistance and aberrant production of apolipoprotein B48 lipoproteins in an animal model of insulin resistance and metabolic dyslipidemia: evidence for activation of protein tyrosine phosphatase-1B, extracellular signal-related kinase, and sterol regulatory element-binding protein-1c in the fructose-fed hamster intestine. Diabetes. 2006;55(5):1316–1326. doi: 10.2337/db04-1084. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

