Methemoglobin and nitric oxide therapy in Ugandan children hospitalized for febrile illness: results from a prospective cohort study and randomized double-blind placebo-controlled trial
- PMID: 27814710
- PMCID: PMC5097382
- DOI: 10.1186/s12887-016-0719-2
Methemoglobin and nitric oxide therapy in Ugandan children hospitalized for febrile illness: results from a prospective cohort study and randomized double-blind placebo-controlled trial
Abstract
Background: Exposure of red blood cells to oxidants increases production of methemoglobin (MHb) resulting in impaired oxygen delivery to tissues. There are no reliable estimates of methemoglobinemia in low resource clinical settings. Our objectives were to: i) evaluate risk factors for methemoglobinemia in Ugandan children hospitalized with fever (study 1); and ii) investigate MHb responses in critically ill Ugandan children with severe malaria treated with inhaled nitric oxide (iNO), an oxidant that induces MHb in a dose-dependent manner (study 2).
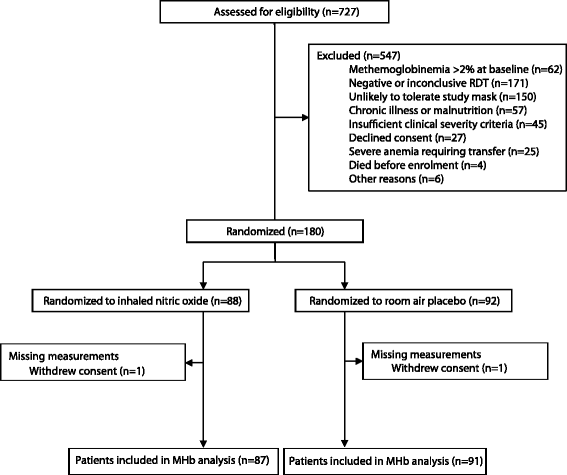
Methods: Two prospective studies were conducted at Jinja Regional Referral Hospital in Uganda between 2011 and 2013. Study 1, a prospective cohort study of children admitted to hospital with fever (fever cohort, n = 2089 children 2 months to 5 years). Study 2, a randomized double-blind placebo-controlled parallel arm trial of room air placebo vs. 80 ppm iNO as an adjunctive therapy for children with severe malaria (RCT, n = 180 children 1-10 years receiving intravenous artesunate and 72 h of study gas). The primary outcomes were: i) masimo pulse co-oximetry elevated MHb levels at admission (>2 %, fever cohort); ii) four hourly MHb levels in the RCT.
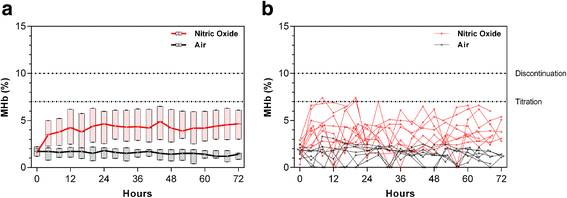
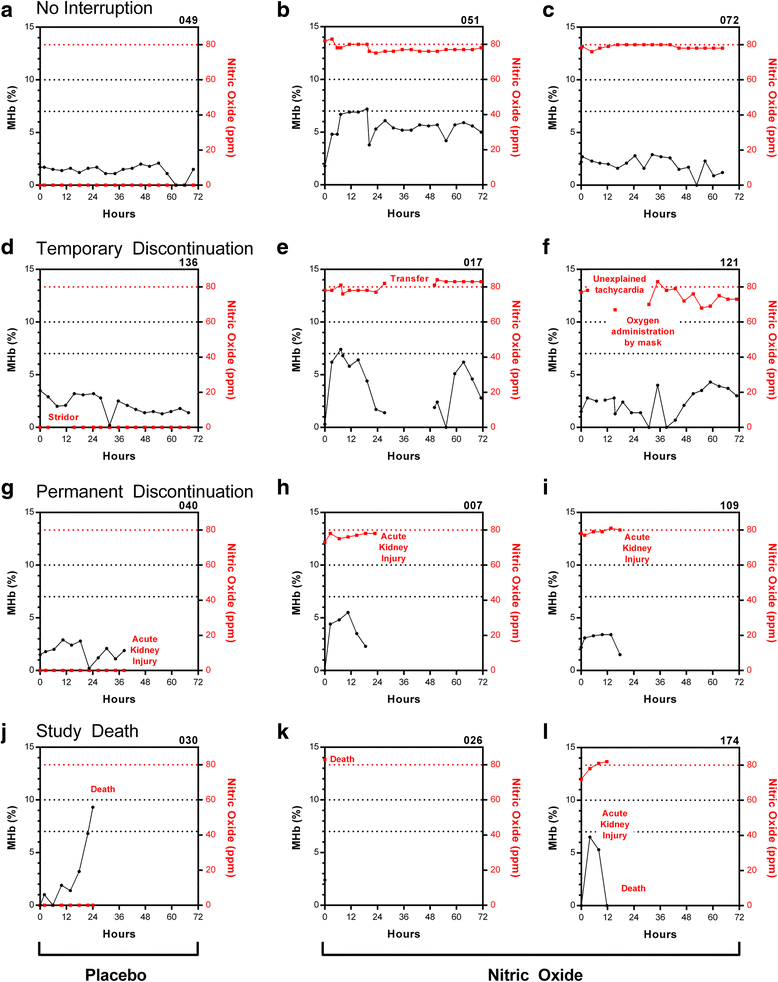
Results: In the fever cohort, 34 % of children admitted with fever had elevated MHb at admission. Children with a history of vomiting, delayed capillary refill, elevated lactate, severe anemia, malaria, or hemoglobinopathies had increased odds of methemoglobinemia (p < 0.05 in a multivariate model). MHb levels at admission were higher in children who died (n = 89) compared to those who survived (n = 1964), p = 0.008. Among children enrolled in the iNO RCT, MHb levels typically plateaued within 12-24 h of starting study gas. MHb levels were higher in children receiving iNO compared to placebo, and MHb > 10 % occurred in 5.7 % of children receiving iNO. There were no differences in rates of study gas discontinuation between trial arms.
Conclusions: Hospitalized children with evidence of impaired oxygen delivery, metabolic acidosis, anemia, or malaria were at risk of methemoglobinemia. However, we demonstrated high-dose iNO could be safely administered to critically ill children with severe malaria with appropriate MHb monitoring.
Trial registration: ClinicalTrials.gov Identifier: NCT01255215 (Date registered: December 5, 2010).
Keywords: Anemia; Fever; Inhaled nitric oxide; Malaria; Metabolic acidosis; Methemoglobin; Oxygen delivery; Pediatrics; Uganda.
Figures
Similar articles
-
Chitinase-3-like 1 is a biomarker of acute kidney injury and mortality in paediatric severe malaria.Malar J. 2018 Feb 15;17(1):82. doi: 10.1186/s12936-018-2225-5. Malar J. 2018. PMID: 29448936 Free PMC article.
-
Inhaled nitric oxide and cognition in pediatric severe malaria: A randomized double-blind placebo controlled trial.PLoS One. 2018 Jan 25;13(1):e0191550. doi: 10.1371/journal.pone.0191550. eCollection 2018. PLoS One. 2018. PMID: 29370261 Free PMC article. Clinical Trial.
-
Inhaled nitric oxide for the adjunctive therapy of severe malaria: protocol for a randomized controlled trial.Trials. 2011 Jul 13;12:176. doi: 10.1186/1745-6215-12-176. Trials. 2011. PMID: 21752262 Free PMC article. Clinical Trial.
-
Inhaled nitric oxide and methemoglobin in full-term infants with persistent pulmonary hypertension of the newborn.Pulm Pharmacol Ther. 2002;15(1):1-5. doi: 10.1006/pupt.2001.0311. Pulm Pharmacol Ther. 2002. PMID: 11969358 Review.
-
Intraoperative detection of methemoglobinemia in a patient given benzocaine spray to relieve discomfort from a nasogastric tube: a case report.AANA J. 2008 Apr;76(2):99-102. AANA J. 2008. PMID: 18478813 Review.
Cited by
-
CD8+ T cells target cerebrovasculature in children with cerebral malaria.J Clin Invest. 2020 Mar 2;130(3):1128-1138. doi: 10.1172/JCI133474. J Clin Invest. 2020. PMID: 31821175 Free PMC article. Clinical Trial.
-
Review 2: Primary graft dysfunction after lung transplant-pathophysiology, clinical considerations and therapeutic targets.J Anesth. 2020 Oct;34(5):729-740. doi: 10.1007/s00540-020-02823-6. Epub 2020 Jul 20. J Anesth. 2020. PMID: 32691226 Free PMC article. Review.
-
Pathogenetic mechanisms and treatment targets in cerebral malaria.Nat Rev Neurol. 2023 Nov;19(11):688-709. doi: 10.1038/s41582-023-00881-4. Epub 2023 Oct 19. Nat Rev Neurol. 2023. PMID: 37857843 Review.
-
Nitric oxide: Clinical applications in critically ill patients.Nitric Oxide. 2022 Apr 1;121:20-33. doi: 10.1016/j.niox.2022.01.007. Epub 2022 Feb 2. Nitric Oxide. 2022. PMID: 35123061 Free PMC article. Review.
-
Chitinase-3-like 1 is a biomarker of acute kidney injury and mortality in paediatric severe malaria.Malar J. 2018 Feb 15;17(1):82. doi: 10.1186/s12936-018-2225-5. Malar J. 2018. PMID: 29448936 Free PMC article.
References
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

