Duration of the anti-androgen in men undergoing 6 months of an LHRH agonist and radiation therapy for unfavorable-risk prostate cancer and the risk of death
- PMID: 27824043
- PMCID: PMC6171772
- DOI: 10.1038/pcan.2016.53
Duration of the anti-androgen in men undergoing 6 months of an LHRH agonist and radiation therapy for unfavorable-risk prostate cancer and the risk of death
Abstract
Background: Whether adding a first-generation anti-androgen (AA) to a luteinizing hormone-releasing hormone agonist in the radiotherapeutic management of unfavorable-risk prostate cancer (PC) reduces the risk of all-cause and PC-specific mortality (ACM and PCSM) among men within differing comorbidity subgroups is unknown.
Methods: Between 1995 and 2001, 206 men with unfavorable-risk PC were enrolled in a randomized trial comparing radiation with or without 6 months of androgen-deprivation therapy (ADT). Partial AA use (median: 4.2 months) occurred in 29 of the 102 men randomized to ADT. Cox, and Fine and Gray's regressions were used to evaluate the impact of full versus partial AA use on PCSM and ACM-risk within comorbidity subgroups.
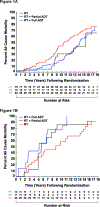
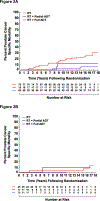
Results: After a median follow-up of 16.62 years, 156 men died. In men with moderate to severe comorbidity increasing death was observed as treatment transitioned from no to partial to full ADT (P=0.02) with an increased ACM-risk with full versus partial AA use (adjusted hazard ratio (AHR), 2.25 (95% confidence interval (CI), 0.94-5.41); P=0.07); whereas only 1 and no PC deaths occurred in men receiving a partial versus full AA course, respectively. Among men with no or minimal comorbidity there was no decrease in ACM (AHR, 0.97 (95% CI, 0.49-1.91); P=0.92) or PCSM-risk (AHR 0.39 (95% CI 0.07-52.18); P=0.28) in comparing full versus partial AA use.
Conclusion: Increasing AA use by 2 months does not appear to impact survival in men with localized unfavorable-risk PC and no or minimal comorbidity, but may shorten survival in men with moderate to severe comorbidity, raising concern regarding in whom and for how long the AA should be prescribed.
Conflict of interest statement
Conflicts of Interest: None
Figures
Similar articles
-
Race and mortality risk after radiation therapy in men treated with or without androgen-suppression therapy for favorable-risk prostate cancer.Cancer. 2016 Dec 1;122(23):3608-3614. doi: 10.1002/cncr.30224. Epub 2016 Aug 4. Cancer. 2016. PMID: 27490845
-
Short-course androgen deprivation therapy and the risk of death from high-risk prostate cancer in men undergoing external beam radiation therapy and brachytherapy.Brachytherapy. 2015 Nov-Dec;14(6):781-7. doi: 10.1016/j.brachy.2015.08.004. Epub 2015 Sep 9. Brachytherapy. 2015. PMID: 26361718
-
The impact of comorbidity and PSA doubling time on the risk of death in men experiencing PSA failure following radiation therapy with or with androgen deprivation therapy for unfavorable-risk prostate cancer.Prostate Cancer Prostatic Dis. 2017 Jun;20(2):234-240. doi: 10.1038/pcan.2016.74. Epub 2017 Jan 24. Prostate Cancer Prostatic Dis. 2017. PMID: 28117382
-
Association of androgen deprivation therapy with cardiovascular death in patients with prostate cancer: a meta-analysis of randomized trials.JAMA. 2011 Dec 7;306(21):2359-66. doi: 10.1001/jama.2011.1745. JAMA. 2011. PMID: 22147380
-
[Androgen deprivation for advanced prostate cancer].Urologe A. 2008 Mar;47(3):270-83. doi: 10.1007/s00120-008-1636-2. Urologe A. 2008. PMID: 18273599 Review. German.
References
-
- Denham JW,Steigler A,Lamb DS, et al. Short term neoadjuvant androgen deprivation and radiotherapy for locally advanced prostate cancer: 10-year data from the TROG 96.01 randomised trial. Lancet Oncol 2011. May;12(5):451–9. - PubMed
-
- D’Amico AV, Manola J, Loffredo M, et al. 6-month androgen suppression plus radiation therapy vs radiation therapy alone for patients with clinically localized prostate cancer: a randomized controlled trial. JAMA 2004. August 18;292(7):821–7. - PubMed
-
- Jones CU, Hunt D, McGowan DG, et al. Radiotherapy and short-term androgen deprivation for localized prostate cancer. N Engl J Med 2011. July 14;365(2):107–18. - PubMed
-
- Bolla M, de Reijke TM, Van Tienhoven G, et al. Duration of androgen suppression in the treatment of prostate cancer. N Engl J Med 2009. June 11;360(24):2516–27. - PubMed
-
- Pilepich MV, Winter K, Lawton CA, et al. Androgen suppression adjuvant to definitive radiotherapy in prostate carcinoma--long-term results of phase III RTOG 85–31. Int J Radiat Oncol Biol Phys 2005. April 1;61(5):1285–90. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

