Invertebrate Iridescent Virus 6, a DNA Virus, Stimulates a Mammalian Innate Immune Response through RIG-I-Like Receptors
- PMID: 27824940
- PMCID: PMC5100955
- DOI: 10.1371/journal.pone.0166088
Invertebrate Iridescent Virus 6, a DNA Virus, Stimulates a Mammalian Innate Immune Response through RIG-I-Like Receptors
Abstract
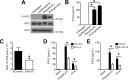
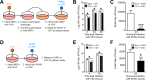
Insects are not only major vectors of mammalian viruses, but are also host to insect-restricted viruses that can potentially be transmitted to mammals. While mammalian innate immune responses to arboviruses are well studied, less is known about how mammalian cells respond to viruses that are restricted to infect only invertebrates. Here we demonstrate that IIV-6, a DNA virus of the family Iridoviridae, is able to induce a type I interferon-dependent antiviral immune response in mammalian cells. Although IIV-6 is a DNA virus, we demonstrate that the immune response activated during IIV-6 infection is mediated by the RIG-I-like receptor (RLR) pathway, and not the canonical DNA sensing pathway via cGAS/STING. We further show that RNA polymerase III is required for maximal IFN-β secretion, suggesting that viral DNA is transcribed by this enzyme into an RNA species capable of activating the RLR pathway. Finally, we demonstrate that the RLR-driven mammalian innate immune response to IIV-6 is functionally capable of protecting cells from subsequent infection with the arboviruses Vesicular Stomatitis virus and Kunjin virus. These results represent a novel example of an invertebrate DNA virus activating a canonically RNA sensing pathway in the mammalian innate immune response, which reduces viral load of ensuing arboviral infection.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
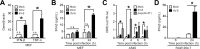




References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

