Prevalence and effect of pre-treatment drug resistance on the virological response to antiretroviral treatment initiated in HIV-infected children - a EuroCoord-CHAIN-EPPICC joint project
- PMID: 27825316
- PMCID: PMC5101717
- DOI: 10.1186/s12879-016-1968-2
Prevalence and effect of pre-treatment drug resistance on the virological response to antiretroviral treatment initiated in HIV-infected children - a EuroCoord-CHAIN-EPPICC joint project
Abstract
Background: Few studies have evaluated the impact of pre-treatment drug resistance (PDR) on response to combination antiretroviral treatment (cART) in children. The objective of this joint EuroCoord-CHAIN-EPPICC/PENTA project was to assess the prevalence of PDR mutations and their association with virological outcome in the first year of cART in children.
Methods: HIV-infected children <18 years initiating cART between 1998 and 2008 were included if having at least one genotypic resistance test prior to cART initiation. We used the World Health Organization 2009 resistance mutation list and Stanford algorithm to infer resistance to prescribed drugs. Time to virological failure (VF) was defined as the first of two consecutive HIV-RNA > 500 copies/mL after 6 months cART and was assessed by Cox proportional hazards models. All models were adjusted for baseline demographic, clinical, immunology and virology characteristics and calendar period of cART start and initial cART regimen.
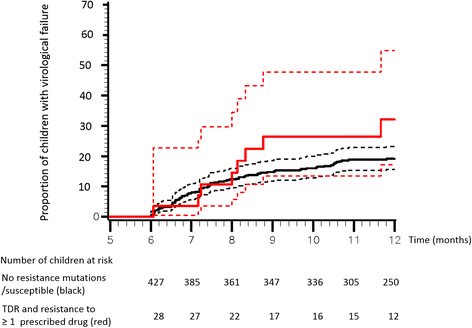
Results: Of 476 children, 88 % were vertically infected. At cART initiation, median (interquartile range) age was 6.6 years (2.1-10.1), CD4 cell count 297 cells/mm3 (98-639), and HIV-RNA 5.2 log10copies/mL (4.7-5.7). Of 37 children (7.8 %, 95 % confidence interval (CI), 5.5-10.6) harboring a virus with ≥1 PDR mutations, 30 children had a virus resistant to ≥1 of the prescribed drugs. Overall, the cumulative Kaplan-Meier estimate for virological failure was 19.8 % (95 %CI, 16.4-23.9). Cumulative risk for VF tended to be higher among children harboring a virus with PDR and resistant to ≥1 drug prescribed than among those receiving fully active cART: 32.1 % (17.2-54.8) versus 19.4 % (15.9-23.6) (P = 0.095). In multivariable analysis, age was associated with a higher risk of VF with a 12 % reduced risk per additional year (HR 0.88; 95 %CI, 0.82-0.95; P < 0.001).
Conclusions: PDR was not significantly associated with a higher risk of VF in children in the first year of cART. The risk of VF decreased by 12 % per additional year at treatment initiation which may be due to fading of PDR mutations over time. Lack of appropriate formulations, in particular for the younger age group, may be an important determinant of virological failure.
Keywords: Children; First-line combination antiretroviral therapy; HIV; Pre-treatment drug resistance mutations; Virological failure.
Figures
Similar articles
-
Effect of transmitted drug resistance on virological and immunological response to initial combination antiretroviral therapy for HIV (EuroCoord-CHAIN joint project): a European multicohort study.Lancet Infect Dis. 2011 May;11(5):363-71. doi: 10.1016/S1473-3099(11)70032-9. Epub 2011 Feb 25. Lancet Infect Dis. 2011. PMID: 21354861 Clinical Trial.
-
Treatment outcomes among HIV-1 and HIV-2 infected children initiating antiretroviral therapy in a concentrated low prevalence setting in West Africa.BMC Pediatr. 2012 Jul 8;12:95. doi: 10.1186/1471-2431-12-95. BMC Pediatr. 2012. PMID: 22770231 Free PMC article. Clinical Trial.
-
Pretreatment HIV drug resistance results in virological failure and accumulation of additional resistance mutations in Ugandan children.J Antimicrob Chemother. 2017 Sep 1;72(9):2587-2595. doi: 10.1093/jac/dkx188. J Antimicrob Chemother. 2017. PMID: 28673027 Free PMC article.
-
Study of the impact of HIV genotypic drug resistance testing on therapy efficacy.Verh K Acad Geneeskd Belg. 2001;63(5):447-73. Verh K Acad Geneeskd Belg. 2001. PMID: 11813503 Review.
-
The association between HIV pretreatment drug resistance and virological outcomes in children and adults in sub-Saharan Africa: A systematic review and meta-analysis.PLoS One. 2024 Apr 16;19(4):e0300456. doi: 10.1371/journal.pone.0300456. eCollection 2024. PLoS One. 2024. PMID: 38626183 Free PMC article.
Cited by
-
AIDS Therapy Evaluation in the Netherlands (ATHENA) national observational HIV cohort: cohort profile.BMJ Open. 2018 Sep 24;8(9):e022516. doi: 10.1136/bmjopen-2018-022516. BMJ Open. 2018. PMID: 30249631 Free PMC article.
-
Clinical Impact of Pretreatment Human Immunodeficiency Virus Drug Resistance in People Initiating Nonnucleoside Reverse Transcriptase Inhibitor-Containing Antiretroviral Therapy: A Systematic Review and Meta-analysis.J Infect Dis. 2021 Aug 2;224(3):377-388. doi: 10.1093/infdis/jiaa683. J Infect Dis. 2021. PMID: 33202025 Free PMC article.
-
Global, regional, and national prevalence of HIV-1 drug resistance in treatment-naive and treatment-experienced children and adolescents: a systematic review and meta-analysis.EClinicalMedicine. 2024 Oct 4;77:102859. doi: 10.1016/j.eclinm.2024.102859. eCollection 2024 Nov. EClinicalMedicine. 2024. PMID: 39430612 Free PMC article.
-
Rates and Correlates of Short Term Virologic Response among Treatment-Naïve HIV-Infected Children Initiating Antiretroviral Therapy in Ethiopia: A Multi-Center Prospective Cohort Study.Pathogens. 2019 Sep 24;8(4):161. doi: 10.3390/pathogens8040161. Pathogens. 2019. PMID: 31554200 Free PMC article.
-
Incidence and predictors of treatment failure among children on first-line antiretroviral therapy in Amhara Region Referral Hospitals, northwest Ethiopia 2018: A retrospective study.PLoS One. 2019 May 1;14(5):e0215300. doi: 10.1371/journal.pone.0215300. eCollection 2019. PLoS One. 2019. PMID: 31042743 Free PMC article.
References
-
- Goodman DD, Zhou Y, Margot NA, McColl DJ, Zhong L, Borroto-Esoda K, Miller MD, Svarovskaia ES. Low level of the K103N HIV-1 above a threshold is associated with virological failure in treatment-naive individuals undergoing efavirenz-containing therapy. AIDS. 2011;25(3):325–333. doi: 10.1097/QAD.0b013e3283427dcb. - DOI - PubMed
-
- Kantor R, Smeaton L, Vardhanabhuti S, Hudelson SE, Wallis CL, Tripathy S, Morgado MG, Saravanan S, Balakrishnan P, Reitsma M, et al. Pretreatment HIV Drug Resistance and HIV-1 Subtype C Are Independently Associated With Virologic Failure: Results From the Multinational PEARLS (ACTG A5175) Clinical Trial. Clin Infect Dis. 2015;60(10):1541-9. - PMC - PubMed
-
- Kuritzkes DR, Lalama CM, Ribaudo HJ, Marcial M, Meyer WA, 3rd, Shikuma C, Johnson VA, Fiscus SA, D’Aquila RT, Schackman BR, et al. Preexisting resistance to nonnucleoside reverse-transcriptase inhibitors predicts virologic failure of an efavirenz-based regimen in treatment-naive HIV-1-infected subjects. J Infect Dis. 2008;197(6):867–870. doi: 10.1086/528802. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

