Interlaboratory Variability in Plasma Creatinine Measurement and the Relation with Estimated Glomerular Filtration Rate and Chronic Kidney Disease Diagnosis
- PMID: 27827312
- PMCID: PMC5220660
- DOI: 10.2215/CJN.05400516
Interlaboratory Variability in Plasma Creatinine Measurement and the Relation with Estimated Glomerular Filtration Rate and Chronic Kidney Disease Diagnosis
Abstract
Background and objectives: The tracing of creatinine (Cr) reference materials to isotope dilution mass spectrometry-assigned values was implemented worldwide to reduce interlaboratory variability and improve assay accuracy. The aims of this study were to examine the current extent of interlaboratory variability and its effect on eGFR.
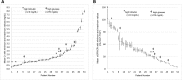
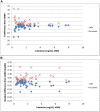
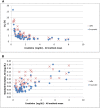
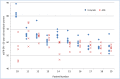
Design, setting, participants, & measurements: Leftover plasma from 2-3 consecutive days was obtained from 53 intensive care unit patients with a range of kidney functions. Individual patient samples were pooled and split and sent to 12 different laboratories for Cr measurement. For each patient, the mean Cr and Chronic Kidney Disease Epidemiology Collaboration eGFR (eGFR-EPI), assuming a 65-year-old nonblack woman, were determined. Interlaboratory variability was assessed by the range and SD of Cr and eGFR-EPI. This was repeated after stratifying by assay type and by the median Cr of 1.36 mg/dl. For patients whose eGFR-EPI range included 60 ml/min per 1.73 m2, the percentage of laboratories with eGFR-EPI<60 ml/min per 1.73 m2 was determined.
Results: The mean±SD of the Cr and eGFR-EPI ranges were 0.20±0.09 mg/dl and 14±9 ml/min per 1.73 m2 for Cr<1.36 mg/dl. Jaffe Cr results were an average 0.1 mg/dl (Cr≥1.36 mg/dl) and 0.05 mg/dl (Cr<1.36 mg/dl) higher than enzymatic results (P<0.001 for both). Ten patients had an eGFR-EPI range that included 60 ml/min per 1.73 m2. Their median eGFR-EPI range was 15 ml/min per 1.73 m2. There was significant discordance in the diagnosis of CKD (eGFR-EPI<60 ml/min per 1.73 m2), with laboratories using Jaffe Cr methods making the diagnosis more frequently than those using enzymatic Cr methods (60% versus 39%).
Conclusions: Significant interlaboratory variability in Cr measurement still exists. Jaffe assays yield higher Cr values than enzymatic assays, leading to lower eGFR-EPIs and more frequent CKD diagnoses. Further improvements in assay performance are required to standardize patient CKD diagnosis and to facilitate longitudinal Cr monitoring across laboratories.
Keywords: African Americans; clinical laboratory techniques; creatinine; enzyme assays; female; glomerular filtration rate; humans; intensive care units; kidney function tests; renal insufficiency, chronic.
Copyright © 2016 by the American Society of Nephrology.
Figures


References
-
- Kidney Disease: Improving Global Outcomes (KDIGO) Work Group : KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int 3: 1–150, 2012 - PubMed
-
- Levey AS: Measurement of renal function in chronic renal disease. Kidney Int 38: 167–184, 1990 - PubMed
-
- Myers GL, Miller WG, Coresh J, Fleming J, Greenberg N, Greene T, Hostetter T, Levey AS, Panteghini M, Welch M, Eckfeldt JH; National Kidney Disease Education Program Laboratory Working Group : Recommendations for improving serum creatinine measurement: a report from the Laboratory Working Group of the National Kidney Disease Education Program. Clin Chem 52: 5–18, 2006 - PubMed
-
- Killeen AA, Ashwood ER, Ventura CB, Styer P: Recent trends in performance and current state of creatinine assays. Arch Pathol Lab Med 137: 496–502, 2013 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

