Randomized, Double-blind, Placebo-controlled Phase III Trial of Duloxetine Monotherapy in Japanese Patients With Chronic Low Back Pain
- PMID: 27831985
- PMCID: PMC5113250
- DOI: 10.1097/BRS.0000000000001707
Randomized, Double-blind, Placebo-controlled Phase III Trial of Duloxetine Monotherapy in Japanese Patients With Chronic Low Back Pain
Abstract
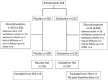
Study design: A 14-week, randomized, double-blind, multicenter, placebo-controlled study of Japanese patients with chronic low back pain (CLBP) who were randomized to either duloxetine 60 mg once daily or placebo.
Objective: This study aimed to assess the efficacy and safety of duloxetine monotherapy in Japanese patients with CLBP.
Summary of background data: In Japan, duloxetine is approved for the treatment of depression, diabetic neuropathic pain, and pain associated with fibromyalgia; however, no clinical study of duloxetine has been conducted for CLBP.
Methods: The primary efficacy measure was the change in the Brief Pain Inventory (BPI) average pain score from baseline to Week 14. Secondary efficacy measures included BPI pain (worst pain, least pain, pain right now), Patient's Global Impression of Improvement, Clinical Global Impressions of Severity, and Roland-Morris Disability Questionnaire, among other measures, and safety and tolerability.
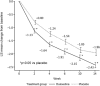
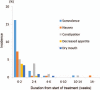
Results: In total, 458 patients were randomized to receive either duloxetine (n = 232) or placebo (n = 226). The BPI average pain score improved significantly in the duloxetine group compared with that in the placebo group at Week 14 [-2.43 ± 0.11 vs. -1.96 ± 0.11, respectively; between-group difference (95% confidence interval), - 0.46 [-0.77 to-0.16]; P = 0.0026]. The duloxetine group showed significant improvement in many secondary measures compared with the placebo group, including BPI pain (least pain, pain right now) (between-group difference: -1.69 ± 0.10, P = 0.0009; -2.42 ± 0.12, P P = 0.0230, respectively), Patient's Global Impression of Improvement (2.46 ± 0.07, P = 0.0026), Clinical Global Impressions of Severity (-1.46 ± 0.06, P = 0.0019), and Roland-Morris Disability Questionnaire (-3.86 ± 0.22, P = 0.0439). Adverse events occurring at a significantly higher incidence in the duloxetine group were somnolence, constipation, nausea, dizziness, and dry mouth, most of which were mild or moderate in severity and were resolved or improved.
Conclusion: Duloxetine 60 mg was effective and well tolerated in Japanese CLBP patients.
Level of evidence: 2.
Figures


References
-
- Yoshimura N, Muraki S, Oka T, et al. Epidemiology of low back pain: from a large scale epidemiologic survey “ROAD”. Nippon Seikei Geka Gakkai Zasshi 2010; 84:437–439.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

