Patient-reported outcome measures of the impact of cancer on patients' everyday lives: a systematic review
- PMID: 27834041
- PMCID: PMC5357497
- DOI: 10.1007/s11764-016-0580-1
Patient-reported outcome measures of the impact of cancer on patients' everyday lives: a systematic review
Abstract
Purpose: Patients with advanced disease are living longer and commonly used patient-reported outcome measures (PROMs) may miss relevant elements of the quality of extended survival. This systematic review examines the measures used to capture aspects of the quality of survival including impact on patients' everyday lives such as finances, work and family roles.
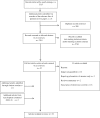
Methods: Searches were conducted in MEDLINE, EMBASE, CINAHL and PsycINFO restricted to English language articles. Information on study characteristics, instruments and outcomes was systematically extracted and synthesised. A predefined set of criteria was used to rate the quality of studies.
Results: From 2761 potentially relevant articles, 22 met all inclusion criteria, including 10 concerning financial distress, 3 on roles and responsibilities and 9 on multiple aspects of social well-being. Generally, studies were not of high quality; many lacked bias free participant selection, had confounding factors and had not accounted for all participants. High levels of financial distress were reported and were associated with multiple demographic factors such as age and income. There were few reports concerned with impacts on patients' roles/responsibilities in everyday life although practical and emotional struggles with parenting were identified. Social difficulties were common and associated with multiple factors including being a caregiver. Many studies were single time-point surveys and used non-validated measures. Exceptions were employment of the COST and Social Difficulties Inventory (SDI), validated measures of financial and social distress respectively.
Conclusions: Impact on some important parts of patients' everyday lives is insufficiently and inconsistently captured. Further PROM development focussing on roles and responsibilities, including work and caring for dependents, is warranted.
Implications for cancer survivors: Factors such as finances, employment and responsibility for caring for dependants (e.g. children and elderly relatives) can affect the well-being of cancer survivors. There is a need to ensure that any instruments used to assess patients' social well-being are broad enough to include these areas so that any difficulties arising can be better understood and appropriately supported.
Keywords: Cancer; Financial burden; Patient-reported outcomes; Quality of life; Quality of survival; Social impact.
Conflict of interest statement
Funding
This review is part of an investigator initiated study funded by Bristol-Myers Squibb.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
As a review paper, this article does not require ethical approval and does not contain any studies with human participants or animals performed by any of the authors.
Figures
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous