Stearoyl-CoA Desaturase-1 Protects Cells against Lipotoxicity-Mediated Apoptosis in Proximal Tubular Cells
- PMID: 27834856
- PMCID: PMC5133868
- DOI: 10.3390/ijms17111868
Stearoyl-CoA Desaturase-1 Protects Cells against Lipotoxicity-Mediated Apoptosis in Proximal Tubular Cells
Abstract
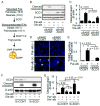
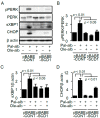
Saturated fatty acid (SFA)-related lipotoxicity is a pathogenesis of diabetes-related renal proximal tubular epithelial cell (PTEC) damage, closely associated with a progressive decline in renal function. This study was designed to identify a free fatty acid (FFA) metabolism-related enzyme that can protect PTECs from SFA-related lipotoxicity. Among several enzymes involved in FFA metabolism, we identified stearoyl-CoA desaturase-1 (SCD1), whose expression level significantly decreased in the kidneys of high-fat diet (HFD)-induced diabetic mice, compared with non-diabetic mice. SCD1 is an enzyme that desaturates SFAs, converting them to monounsaturated fatty acids (MUFAs), leading to the formation of neutral lipid droplets. In culture, retrovirus-mediated overexpression of SCD1 or MUFA treatment significantly ameliorated SFA-induced apoptosis in PTECs by enhancing intracellular lipid droplet formation. In contrast, siRNA against SCD1 exacerbated the apoptosis. Both overexpression of SCD1 and MUFA treatment reduced SFA-induced apoptosis via reducing endoplasmic reticulum stress in cultured PTECs. Thus, HFD-induced decrease in renal SCD1 expression may play a pathogenic role in lipotoxicity-induced renal injury, and enhancing SCD1-mediated desaturation of SFA and subsequent formation of neutral lipid droplets may become a promising therapeutic target to reduce SFA-induced lipotoxicity. The present study provides a novel insight into lipotoxicity in the pathogenesis of diabetic nephropathy.
Keywords: desaturation; free fatty acid; lipid droplet; lipotoxicity; proximal tubular epithelial cells.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Diabetes Control and Complications Trial Research Group The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. The diabetes control and complications trial research group. N. Engl. J. Med. 1993;329:977–986. - PubMed
-
- Brenner B.M., Cooper M.E., de Zeeuw D., Keane W.F., Mitch W.E., Parving H.H., Remuzzi G., Snapinn S.M., Zhang Z., Shahinfar S., et al. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N. Engl. J. Med. 2001;345:861–869. doi: 10.1056/NEJMoa011161. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

