Sustained Delivery of Timolol Maleate for Over 90 Days by Subconjunctival Injection
- PMID: 27835065
- PMCID: PMC5165680
- DOI: 10.1089/jop.2016.0042
Sustained Delivery of Timolol Maleate for Over 90 Days by Subconjunctival Injection
Abstract
Purpose: Medical treatment of glaucoma relies on intraocular pressure (IOP)-lowering medications, typically administered daily by the patient. While these medications are effective when applied correctly, patient adherence is a major obstacle in glaucoma treatment. We have developed a sustained-release formulation of timolol maleate that can be injected subconjunctivally to avoid patient noncompliance.
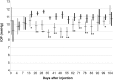
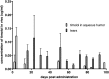
Methods: A biodegradable microsphere formulation for timolol maleate was injected subconjunctivally in normal rabbits. We measured timolol levels in tears, aqueous humor, vitreous humor, and serum of study rabbits. Furthermore, IOP profiles were recorded longitudinally. Tissue compatibility and side effects were evaluated using histochemistry.
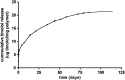
Results: The microsphere formulation led to measureable amounts of timolol in the aqueous humor and the tear film for up to 90 days. Timolol was not detectable in the serum at any time. A significant reduction of IOP was observed in treated eyes. Clinically, the subconjunctival administration of the microspheres was well tolerated with no signs of inflammation or infection. The absence of local inflammation was confirmed by histology.
Conclusions: A single subconjunctival administration of timolol microspheres achieved delivery and IOP reduction in rabbits for up to 90 days without local or systemic inflammation or toxicity. This approach has the potential to improve the management of glaucoma in patient populations, who are challenged to adhere to a regimen of daily eye drops.
Keywords: glaucoma pharmacology; rabbit; sustained delivery; timolol maleate.
Conflict of interest statement
Author Disclosure Statement No competing financial interests exist.
Figures


References
-
- Oliver J.E., Hattenhauer M.G., Herman D., Hodge D.O., Kennedy R., Fang-Yen M., and Johnson D.H. Blindness and glaucoma: a comparison of patients progressing to blindness from glaucoma with patients maintaining vision. Am. J. Ophthalmol. 133:764–772, 2002 - PubMed
-
- Kuehn M.H., Fingert J.H., and Kwon Y.H. Retinal ganglion cell death in glaucoma: mechanisms and neuroprotective strategies. Ophthalmol. Clin. North Am. 18:383–395, vi, 2005 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

