Extracellular Vesicles Isolated from the Leaf Apoplast Carry Stress-Response Proteins
- PMID: 27837092
- PMCID: PMC5210723
- DOI: 10.1104/pp.16.01253
Extracellular Vesicles Isolated from the Leaf Apoplast Carry Stress-Response Proteins
Abstract
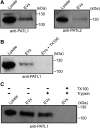
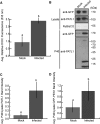
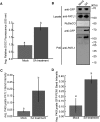
Exosomes are extracellular vesicles (EVs) that play a central role in intercellular signaling in mammals by transporting proteins and small RNAs. Plants are also known to produce EVs, particularly in response to pathogen infection. The contents of plant EVs have not been analyzed, however, and their function is unknown. Here, we describe a method for purifying EVs from the apoplastic fluids of Arabidopsis (Arabidopsis thaliana) leaves. Proteomic analyses of these EVs revealed that they are highly enriched in proteins involved in biotic and abiotic stress responses. Consistent with this finding, EV secretion was enhanced in plants infected with Pseudomonas syringae and in response to treatment with salicylic acid. These findings suggest that EVs may represent an important component of plant immune responses.
© 2017 American Society of Plant Biologists. All Rights Reserved.
Figures
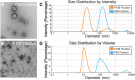

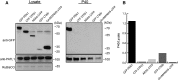
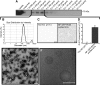
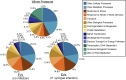
References
-
- Admyre C, Johansson SM, Qazi KR, Filén JJ, Lahesmaa R, Norman M, Neve EP, Scheynius A, Gabrielsson S (2007) Exosomes with immune modulatory features are present in human breast milk. J Immunol 179: 1969–1978 - PubMed
-
- Agrawal GK, Jwa NS, Lebrun MH, Job D, Rakwal R (2010) Plant secretome: unlocking secrets of the secreted proteins. Proteomics 10: 799–827 - PubMed
-
- Alexandersson E, Saalbach G, Larsson C, Kjellbom P (2004) Arabidopsis plasma membrane proteomics identifies components of transport, signal transduction and membrane trafficking. Plant Cell Physiol 45: 1543–1556 - PubMed

