Tumor Reduction in Primary and Metastatic Pancreatic Cancer Lesions With nab-Paclitaxel and Gemcitabine: An Exploratory Analysis From a Phase 3 Study
- PMID: 27841795
- PMCID: PMC5266413
- DOI: 10.1097/MPA.0000000000000742
Tumor Reduction in Primary and Metastatic Pancreatic Cancer Lesions With nab-Paclitaxel and Gemcitabine: An Exploratory Analysis From a Phase 3 Study
Abstract
Objectives: Results from the phase 3 Metastatic Pancreatic Adenocarcinoma Clinical Trial (MPACT) led to approval of nab-paclitaxel plus gemcitabine for first-line treatment of metastatic pancreatic cancer. The current analysis evaluated the effects of nab-paclitaxel plus gemcitabine versus gemcitabine on primary pancreatic and metastatic lesions.
Methods: In this analysis of the previously described MPACT trial, changes in pancreatic and metastatic tumor burden were assessed using independently measured diameters of lesions on computed tomography or magnetic resonance imaging scans. Changes in the sums of longest tumor diameters were summarized using descriptive statistics and were included in a multivariate analysis of overall survival.
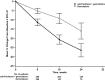
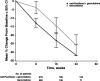
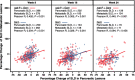
Results: Primary pancreatic lesion measurement was feasible. Reductions in primary pancreatic tumor burden and metastatic burden from baseline to nadir were significantly greater with nab-paclitaxel plus gemcitabine versus gemcitabine. Baseline pancreatic tumor burden was independently predictive of survival. Both regimens elicited linear reductions in primary pancreatic and metastatic tumor burden through time. There was a high within-patient concordance of tumor changes between primary pancreatic lesions and metastatic lesions.
Conclusions: This analysis of MPACT demonstrated significant tumor shrinkage benefit for nab-paclitaxel plus gemcitabine in both primary pancreatic and metastatic lesions, supporting ongoing evaluation of this regimen in locally advanced disease.
Figures
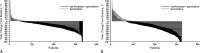
References
-
- National Cancer Institute-Surveillance, Epidemiology, and End Results Program. SEER stat fact sheets: pancreas cancer. Available at: http://seer.cancer.gov/statfacts/html/pancreas.html. Accessed June 17, 2016.
-
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA Cancer J Clin. 2015;65:5–29. - PubMed
-
- Ducreux M, Cuhna AS, Caramella C, et al. Cancer of the pancreas: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2015;26(suppl 5):v56–v68. - PubMed
-
- National Comprehensive Cancer Network. Clinical practice guidelines in oncology. Pancreatic adenocarcinoma. V1. 2016. Available at: https://www.nccn.org/professionals/physician_gls/pdf/pancreatic.pdf. Accessed June 17, 2016.
-
- American Cancer Society. Cancer facts and figures. 2016. Available at: http://www.cancer.org/acs/groups/content/@research/documents/document/ac.... Accessed June 17, 2016.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

