In vivo characterization of distinct modality-specific subsets of somatosensory neurons using GCaMP
- PMID: 27847865
- PMCID: PMC5106201
- DOI: 10.1126/sciadv.1600990
In vivo characterization of distinct modality-specific subsets of somatosensory neurons using GCaMP
Abstract
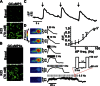
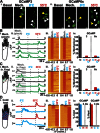
Mechanistic insights into pain pathways are essential for a rational approach to treating this vast and increasing clinical problem. Sensory neurons that respond to tissue damage (nociceptors) may evoke pain sensations and are typically classified on the basis of action potential velocity. Electrophysiological studies have suggested that most of the C-fiber nociceptors are polymodal, responding to a variety of insults. In contrast, gene deletion studies in the sensory neurons of transgenic mice have frequently resulted in modality-specific deficits. We have used an in vivo imaging approach using the genetically encoded fluorescent calcium indicator GCaMP to study the activity of dorsal root ganglion sensory neurons in live animals challenged with painful stimuli. Using this approach, we can visualize spatially distinct neuronal responses and find that >85% of responsive dorsal root ganglion neurons are modality-specific, responding to either noxious mechanical, cold, or heat stimuli. These observations are mirrored in behavioral studies of transgenic mice. For example, deleting sodium channel Nav1.8 silences mechanical- but not heat-sensing sensory neurons, consistent with behavioral deficits. In contrast, primary cultures of axotomized sensory neurons show high levels of polymodality. After intraplantar treatment with prostaglandin E2, neurons in vivo respond more intensely to noxious thermal and mechanical stimuli, and additional neurons (silent nociceptors) are unmasked. Together, these studies define polymodality as an infrequent feature of nociceptive neurons in normal animals.
Keywords: GCaMP imaging; Pain modality; nociception; polymodal nociceptor.
Figures




References
-
- Davis J. B., Gray J., Gunthorpe M. J., Hatcher J. P., Davey P. T., Overend P., Harries M. H., Latcham J., Clapham C., Atkinson K., Hughes S. A., Rance K., Grau E., Harper A. J., Pugh P. L., Rogers D. C., Bingham S., Randall A., Sheardown S. A., Vanilloid receptor-1 is essential for inflammatory thermal hyperalgesia. Nature 405, 183–187 (2000). - PubMed
-
- Gasser H. S., The classification of nerve fibers. Ohio J. Sci. 41, 145–159 (1941).
-
- Djouhri L., Lawson S. N., Aβ-fiber nociceptive primary afferent neurons: A review of incidence and properties in relation to other afferent A-fiber neurons in mammals. Brain Res. Rev. 46, 131–145 (2004). - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

