Speeding Access to Vaccines and Medicines in Low- and Middle-Income Countries: A Case for Change and a Framework for Optimized Product Market Authorization
- PMID: 27851831
- PMCID: PMC5112794
- DOI: 10.1371/journal.pone.0166515
Speeding Access to Vaccines and Medicines in Low- and Middle-Income Countries: A Case for Change and a Framework for Optimized Product Market Authorization
Abstract
Background: The United Nations Millennium Development Goals galvanized global efforts to alleviate suffering of the world's poorest people through unprecedented public-private partnerships. Donor aid agencies have demonstrably saved millions of lives that might otherwise have been lost to disease through increased access to quality-assured vaccines and medicines. Yet, the introduction of these health interventions in low- and middle-income countries (LMICs) continues to face a time lag due to factors which remain poorly understood.
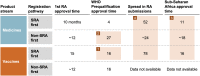
Methods and findings: A recurring theme from our partnership engagements was that an optimized regulatory process would contribute to improved access to quality health products. Therefore, we investigated the current system for medicine and vaccine registration in LMICs as part of our comprehensive regulatory strategy. Here, we report a fact base of the registration timelines for vaccines and drugs used to treat certain communicable diseases in LMICs. We worked with a broad set of stakeholders, including the World Health Organization's prequalification team, national regulatory authorities, manufacturers, procurers, and other experts, and collected data on the timelines between first submission and last approval of applications for product registration sub-Saharan Africa. We focused on countries with the highest burden of communicable disease and the greatest need for the products studied. The data showed a typical lag of 4 to 7 years between the first regulatory submission which was usually to a regulatory agency in a high-income country, and the final approval in Sub-Saharan Africa. Two of the three typical registration steps which products undergo before delivery in the countries involve lengthy timelines. Failure to leverage or rely on the findings from reviews already performed by competent regulatory authorities, disparate requirements for product approval by the countries, and lengthy timelines by manufacturers to respond to regulatory queries were key underlying factors for the delays.
Conclusions: We propose a series of measures which we developed in close collaboration with key stakeholders that could be taken to reduce registration time and to make safe, effective medicines more quickly available in countries where they are most needed. Many of these recommendations are being implemented by the responsible stakeholders, including the WHO prequalification team and the national regulatory authorities in Sub-Saharan Africa. Those efforts will be the focus of subsequent publications by the pertinent groups.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


References
-
- United Nations Millennium Development Goals. Available: http://www.un.org/millenniumgoals/bkgd.shtml. Accessed 1 April 2015.
-
- GAVI Alliance. Global level indicators. 2015, April.Available: http://www.gavialliance.org/results/gavi_alliance_goal_level_indicators/ Accessed 27 April, 2015.
-
- Goosby E, Dybul M, Fauci AS, Fu J, Walsh T, Needle R, et al. The United States President's Emergency Plan for AIDS Relief: a story of partnerships and smart investments to turn the tide of the global AIDS pandemic. J Acquir Immune Defic Syndr. 2012. August 15;60 Suppl 3:S51–6. 10.1097/QAI.0b013e31825ca721 - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

