Culture expansion of adipose derived stromal cells. A closed automated Quantum Cell Expansion System compared with manual flask-based culture
- PMID: 27852267
- PMCID: PMC5112664
- DOI: 10.1186/s12967-016-1080-9
Culture expansion of adipose derived stromal cells. A closed automated Quantum Cell Expansion System compared with manual flask-based culture
Abstract
Background: Adipose derived stromal cells (ASCs) are a rich and convenient source of cells for clinical regenerative therapeutic approaches. However, applications of ASCs often require cell expansion to reach the needed dose. In this study, cultivation of ASCs from stromal vascular fraction (SVF) over two passages in the automated and functionally closed Quantum Cell Expansion System (Quantum system) is compared with traditional manual cultivation.
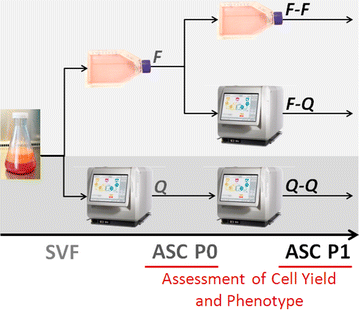
Methods: Stromal vascular fraction was isolated from abdominal fat, suspended in α-MEM supplemented with 10% Fetal Bovine Serum and seeded into either T75 flasks or a Quantum system that had been coated with cryoprecipitate. The cultivation of ASCs from SVF was performed in 3 ways: flask to flask; flask to Quantum system; and Quantum system to Quantum system. In all cases, quality controls were conducted for sterility, mycoplasmas, and endotoxins, in addition to the assessment of cell counts, viability, immunophenotype, and differentiation potential.
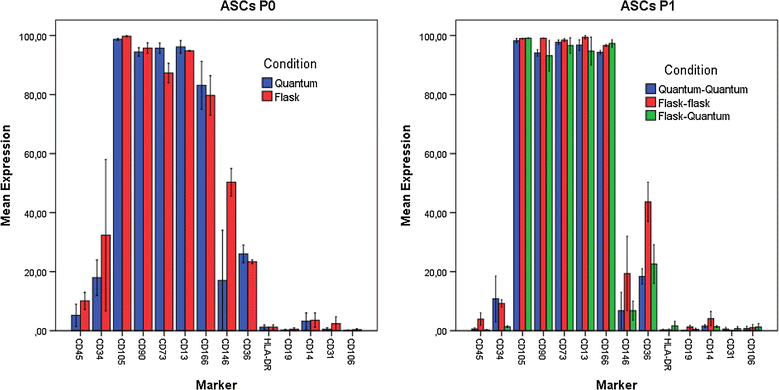
Results: The viability of ASCs passage 0 (P0) and P1 was above 96%, regardless of cultivation in flasks or Quantum system. Expression of surface markers and differentiation potential was consistent with ISCT/IFATS standards for the ASC phenotype. Sterility, mycoplasma, and endotoxin tests were consistently negative. An average of 8.0 × 107 SVF cells loaded into a Quantum system yielded 8.96 × 107 ASCs P0, while 4.5 × 106 SVF cells seeded per T75 flask yielded an average of 2.37 × 106 ASCs-less than the number of SVF cells seeded. ASCs P1 expanded in the Quantum system demonstrated a population doubling (PD) around 2.2 regardless of whether P0 was previously cultured in flasks or Quantum, while ASCs P1 in flasks only reached a PD of 1.0.
Conclusion: Manufacturing of ASCs in a Quantum system enhances ASC expansion rate and yield significantly relative to manual processing in T-flasks, while maintaining the purity and quality essential to safe and robust cell production. Notably, the use of the Quantum system entails significantly reduced working hours and thereby costs.
Keywords: Adipose derived stromal cells; Bioreactor; Cell culture; Cell expansion; Clinical application; Coating; Cryoprecipitate; Mesenchymal stem cell; Storage.
Figures

Similar articles
-
Development of large-scale manufacturing of adipose-derived stromal cells for clinical applications using bioreactors and human platelet lysate.Scand J Clin Lab Invest. 2018 Jul;78(4):293-300. doi: 10.1080/00365513.2018.1462082. Epub 2018 Apr 17. Scand J Clin Lab Invest. 2018. PMID: 29661028
-
Clinical-scale expansion of adipose-derived stromal cells starting from stromal vascular fraction in a single-use bioreactor: proof of concept for autologous applications.J Tissue Eng Regen Med. 2018 Jan;12(1):129-141. doi: 10.1002/term.2377. Epub 2017 May 12. J Tissue Eng Regen Med. 2018. PMID: 27943660
-
Immunophenotype of human adipose-derived cells: temporal changes in stromal-associated and stem cell-associated markers.Stem Cells. 2006 Feb;24(2):376-85. doi: 10.1634/stemcells.2005-0234. Epub 2005 Dec 1. Stem Cells. 2006. PMID: 16322640
-
Human Adipose Stem Cells: From Bench to Bedside.Tissue Eng Part B Rev. 2015 Dec;21(6):572-84. doi: 10.1089/ten.TEB.2014.0608. Epub 2015 Jun 17. Tissue Eng Part B Rev. 2015. PMID: 25953464 Review.
-
Revisiting the Advances in Isolation, Characterization and Secretome of Adipose-Derived Stromal/Stem Cells.Int J Mol Sci. 2018 Jul 27;19(8):2200. doi: 10.3390/ijms19082200. Int J Mol Sci. 2018. PMID: 30060511 Free PMC article. Review.
Cited by
-
Membrane Applications in Autologous Cell Therapy.Membranes (Basel). 2022 Nov 24;12(12):1182. doi: 10.3390/membranes12121182. Membranes (Basel). 2022. PMID: 36557091 Free PMC article. Review.
-
GMP Compliant Production of a Cryopreserved Adipose-Derived Stromal Cell Product for Feasible and Allogeneic Clinical Use.Stem Cells Int. 2022 Jun 20;2022:4664917. doi: 10.1155/2022/4664917. eCollection 2022. Stem Cells Int. 2022. PMID: 35769340 Free PMC article.
-
Laminin as a Potent Substrate for Large-Scale Expansion of Human Induced Pluripotent Stem Cells in a Closed Cell Expansion System.Stem Cells Int. 2019 Jan 22;2019:9704945. doi: 10.1155/2019/9704945. eCollection 2019. Stem Cells Int. 2019. PMID: 30805013 Free PMC article.
-
Three-Dimensional Bioreactor Technologies for the Cocultivation of Human Mesenchymal Stem/Stromal Cells and Beta Cells.Stem Cells Int. 2018 Mar 14;2018:2547098. doi: 10.1155/2018/2547098. eCollection 2018. Stem Cells Int. 2018. PMID: 29731775 Free PMC article. Review.
-
GMP Production and Scale-Up of Adherent Neural Stem Cells with a Quantum Cell Expansion System.Mol Ther Methods Clin Dev. 2018 Jul 7;10:48-56. doi: 10.1016/j.omtm.2018.05.006. eCollection 2018 Sep 21. Mol Ther Methods Clin Dev. 2018. PMID: 29992178 Free PMC article.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

