A viscoelastic-stochastic model of the effects of cytoskeleton remodelling on cell adhesion
- PMID: 27853571
- PMCID: PMC5098996
- DOI: 10.1098/rsos.160539
A viscoelastic-stochastic model of the effects of cytoskeleton remodelling on cell adhesion
Abstract
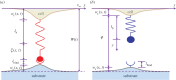
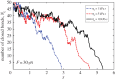
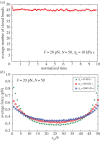
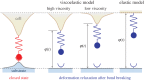
Cells can adapt their mechanical properties through cytoskeleton remodelling in response to external stimuli when the cells adhere to the extracellular matrix (ECM). Many studies have investigated the effects of cell and ECM elasticity on cell adhesion. However, experiments determined that cells are viscoelastic and exhibiting stress relaxation, and the mechanism behind the effect of cellular viscoelasticity on the cell adhesion behaviour remains unclear. Therefore, we propose a theoretical model of a cluster of ligand-receptor bonds between two dissimilar viscoelastic media subjected to an applied tensile load. In this model, the distribution of interfacial traction is assumed to follow classical continuum viscoelastic equations, whereas the rupture and rebinding of individual molecular bonds are governed by stochastic equations. On the basis of this model, we determined that viscosity can significantly increase the lifetime, stability and dynamic strength of the adhesion cluster of molecular bonds, because deformation relaxation attributed to the viscoelastic property can increase the rebinding probability of each open bond and reduce the stress concentration in the adhesion area.
Keywords: Monte Carlo simulation; cell adhesion; viscoelastic–stochastic model.
Figures



References
-
- Lo CM, Wang HB, Dembo M, Wang YL. 2000. Cell movement is guided by the rigidity of the substrate. Biophys. J. 79, 144–152. (doi:10.1016/S0006-3495(00)76279-5) - DOI - PMC - PubMed
-
- Discher DE, Janmey P, Wang YL. 2005. Tissue cells feel and respond to the stiffness of their substrate. Science 310, 1139–1143. (doi:10.1126/science.1116995) - DOI - PubMed
-
- Peyton SR, Putnam AJ. 2005. Extracellular matrix rigidity governs smooth muscle cell motility in a biphasic fashion. J. Cell. Physiol. 204, 198–209. (doi:10.1002/jcp.20274) - DOI - PubMed
-
- Zhong Y, Ji B. 2014. How do cells produce and regulate the driving force in the process of migration? Eur. Phys. J. Spec. Top. 223, 1373–1390. (doi:10.1140/epjst/e2014-02196-8) - DOI
-
- Engler AJ, Richert L, Wong JY, Picart C, Discher DE. 2004. Surface probe measurements of the elasticity of sectioned tissue, thin gels and polyelectrolyte multilayer films: correlations between substrate stiffness and cell adhesion. Surf. Sci. 570, 142–154. (doi:10.1016/j.susc.2004.06.179) - DOI
LinkOut - more resources
Full Text Sources
Other Literature Sources
