Metabolism and phospholipid assembly of polyunsaturated fatty acids in human bone marrow mesenchymal stromal cells
- PMID: 27856675
- PMCID: PMC5234713
- DOI: 10.1194/jlr.M070680
Metabolism and phospholipid assembly of polyunsaturated fatty acids in human bone marrow mesenchymal stromal cells
Abstract
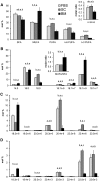
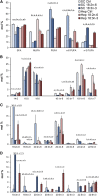
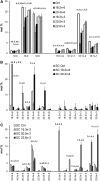
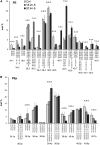
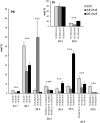
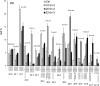
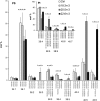
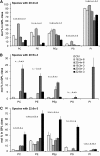
High arachidonic acid (20:4n-6) and low n-3 PUFA levels impair the capacity of cultured human bone marrow mesenchymal stromal cells (hBMSCs) to modulate immune functions. The capacity of the hBMSCs to modify PUFA structures was found to be limited. Therefore, different PUFA supplements given to the cells resulted in very different glycerophospholipid (GPL) species profiles and substrate availability for phospholipases, which have preferences for polar head group and acyl chains when liberating PUFA precursors for production of lipid mediators. When supplemented with 20:4n-6, the cells increased prostaglandin E2 secretion. However, they elongated 20:4n-6 to the less active precursor, 22:4n-6, and also incorporated it into triacylglycerols, which may have limited the proinflammatory signaling. The n-3 PUFA precursor, 18:3n-3, had little potency to reduce the GPL 20:4n-6 content, while the eicosapentaenoic (20:5n-3) and docosahexaenoic (22:6n-3) acid supplements efficiently displaced the 20:4n-6 acyls, and created diverse GPL species substrate pools allowing attenuation of inflammatory signaling. The results emphasize the importance of choosing appropriate PUFA supplements for in vitro hBMSC expansion and suggests that for optimal function they require an exogenous fatty acid source providing 20:5n-3 and 22:6n-3 sufficiently, but 20:4n-6 moderately, which calls for specifically designed optimal PUFA supplements for the cultures.
Keywords: arachidonic acid; docosahexaenoic acid; eicosapentaenoic acid; glycerophospholipid; immunomodulation; lipid signaling; mass spectrometry; mesenchymal stromal/stem cell; prostaglandin E2.
Copyright © 2017 by the American Society for Biochemistry and Molecular Biology, Inc.
Figures




References
-
- Pittenger M. F., Mackay A. M., Beck S. C., Jaiswal R. K., Douglas R., Mosca J. D., Moorman M. A., Simonetti D. W., Craig S., and Marshak D. R.. 1999. Multilineage potential of adult human mesenchymal stem cells. Science. 284: 143–147. - PubMed
-
- Di Nicola M., Carlo-Stella C., Magni M., Milanesi M., Longoni P. D., Matteucci P., Grisanti S., and Gianni A. M.. 2002. Human bone marrow stromal cells suppress T-lymphocyte proliferation induced by cellular or nonspecific mitogenic stimuli. Blood. 99: 3838–3843. - PubMed
-
- Sato K., Ozaki K., Oh I., Meguro A., Hatanaka K., Nagai T., Muroi K., and Ozawa K.. 2007. Nitric oxide plays a critical role in suppression of T-cell proliferation by mesenchymal stem cells. Blood. 109: 228–234. - PubMed
-
- Ren G., Zhang L., Zhao X., Xu G., Zhang Y., Roberts A. I., Zhao R. C. C., and Shi Y.. 2008. Mesenchymal stem cell-mediated immunosuppression occurs via concerted action of chemokines and nitric oxide. Cell Stem Cell. 2: 141–150. - PubMed
-
- Krampera M., Glennie S., Dyson J., Scott D., Laylor R., Simpson E., and Dazzi F.. 2003. Bone marrow mesenchymal stem cells inhibit the response of naive and memory antigen-specific T cells to their cognate peptide. Blood. 101: 3722–3729. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

