Some Like It Hot: Heat Resistance of Escherichia coli in Food
- PMID: 27857712
- PMCID: PMC5093140
- DOI: 10.3389/fmicb.2016.01763
Some Like It Hot: Heat Resistance of Escherichia coli in Food
Abstract
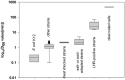
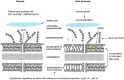
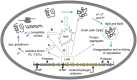
Heat treatment and cooking are common interventions for reducing the numbers of vegetative cells and eliminating pathogenic microorganisms in food. Current cooking method requires the internal temperature of beef patties to reach 71°C. However, some pathogenic Escherichia coli such as the beef isolate E. coli AW 1.7 are extremely heat resistant, questioning its inactivation by current heat interventions in beef processing. To optimize the conditions of heat treatment for effective decontaminations of pathogenic E. coli strains, sufficient estimations, and explanations are necessary on mechanisms of heat resistance of target strains. The heat resistance of E. coli depends on the variability of strains and properties of food formulations including salt and water activity. Heat induces alterations of E. coli cells including membrane, cytoplasm, ribosome and DNA, particularly on proteins including protein misfolding and aggregations. Resistant systems of E. coli act against these alterations, mainly through gene regulations of heat response including EvgA, heat shock proteins, σE and σS, to re-fold of misfolded proteins, and achieve antagonism to heat stress. Heat resistance can also be increased by expression of key proteins of membrane and stabilization of membrane fluidity. In addition to the contributions of the outer membrane porin NmpC and overcome of osmotic stress from compatible solutes, the new identified genomic island locus of heat resistant performs a critical role to these highly heat resistant strains. This review aims to provide an overview of current knowledge on heat resistance of E. coli, to better understand its related mechanisms and explore more effective applications of heat interventions in food industry.
Keywords: Escherichia coli; VTEC; food processing; heat resistance; locus of heat resistance; protein.
Figures
References
-
- Ahmed N. M., Conner D. E., Huffman D. L. (1995). Heat resistance of Escherichia coli O157:H7 in meat and poultry as affected by product composition. J. Food Sci. 60 606–610. 10.1111/j.1365-2621.1995.tb09838.x - DOI
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

