Effects of biological tissue structural anisotropy and anisotropy of magnetic susceptibility on the gradient echo MRI signal phase: theoretical background
- PMID: 27862452
- PMCID: PMC6375105
- DOI: 10.1002/nbm.3655
Effects of biological tissue structural anisotropy and anisotropy of magnetic susceptibility on the gradient echo MRI signal phase: theoretical background
Abstract
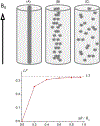
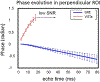
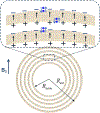
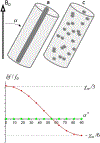
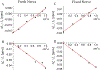
Quantitative susceptibility mapping is a potentially powerful technique for mapping tissue magnetic susceptibility from gradient recalled echo (GRE) MRI signal phase. In this review, we present up-to-date theoretical developments in analyzing the relationships between GRE signal phase and the underlying tissue microstructure and magnetic susceptibility at the cellular level. Two important phenomena contributing to the GRE signal phase are at the focus of this review - tissue structural anisotropy (e.g. cylindrical axonal bundles in white matter) and magnetic susceptibility anisotropy. One of the most intriguing and challenging problems in this field is calculating the so-called Lorentzian contribution to the phase shift induced by the local environment - magnetized tissue structures that have dimensions smaller than the imaging voxel (e.g. cells, cellular components, blood capillaries). In this review, we briefly discuss a "standard" approach to this problem, based on introduction of an imaginary Lorentzian cavity, as well as a more recent method - the generalized Lorentzian tensor approach (GLTA) - that is based on a statistical approach and a direct solution of the magnetostatic Maxwell equations. The latter adequately accounts for both types of anisotropy: the anisotropy of magnetic susceptibility and the structural tissue anisotropy. In the GLTA the frequency shift due to the local environment is characterized by the Lorentzian tensor L^, which has a substantially different structure than the susceptibility tensor χ^. While the components of χ^ are compartmental susceptibilities "weighted" by their volume fractions, the components of L^ are weighted by specific numerical factors depending on tissue geometrical microsymmetry. In multi-compartment structures, the components of the Lorentzian tensor also depend on the compartmental relaxation properties, hence the MR pulse sequence settings. Copyright © 2016 John Wiley & Sons, Ltd.
Keywords: generalized Lorentzian tensor approach; magnetic susceptibility; phase contrast; quantitative susceptibility mapping; white matter.
Copyright © 2016 John Wiley & Sons, Ltd.
Figures



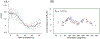

References
-
- Zhong K, Leupold J, von Elverfeldt D, Speck O. The molecular basis for gray and white matter contrast in phase imaging. Neuroimage 2008; 40(4): 1561–1566. - PubMed
-
- Marques JP, Maddage R, Mlynarik V, Gruetter R. On the origin of the MR image phase contrast: an in vivo MR microscopy study of the rat brain at 14.1 T. Neuroimage 2009; 46(2): 345–352. - PubMed
-
- Abduljalil AM, Schmalbrock P, Novak V, Chakeres DW. Enhanced gray and white matter contrast of phase susceptibility-weighted images in ultra-high-field magnetic resonance imaging. J. Magn. Reson. Imaging 2003; 18(3): 284–290. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

