Effects of covalent modification by 4-hydroxy-2-nonenal on the noncovalent oligomerization of ubiquitin
- PMID: 27862610
- PMCID: PMC5360464
- DOI: 10.1002/jms.3897
Effects of covalent modification by 4-hydroxy-2-nonenal on the noncovalent oligomerization of ubiquitin
Abstract
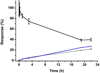
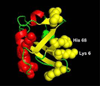
When lipid membranes containing ω-6 polyunsaturated fatty acyl chains are subjected to oxidative stress, one of the reaction products is 4-hydroxy-2-nonenal (HNE)-a chemically reactive short chain alkenal that can covalently modify proteins. The ubiquitin proteasome system is involved in the clearing of proteins modified by oxidation products such as HNE, but the chemical structure, stability and function of ubiquitin may be impaired by HNE modification. To evaluate this possibility, the susceptibility of ubiquitin to modification by HNE has been characterized over a range of concentrations where ubiquitin forms non-covalent oligomers. Results indicate that HNE modifies ubiquitin at only two of the many possible sites, and that HNE modification at these two sites alters the ubiquitin oligomerization equilibrium. These results suggest that any role ubiquitin may have in clearing proteins damaged by oxidative stress may itself be impaired by oxidative lipid degradation products. Copyright © 2016 John Wiley & Sons, Ltd.
Keywords: 4-hydroxy-2-nonenal; Alzheimer's disease; oxidative stress; photocrosslinking; tandem mass spectrometry; ubiquitin.
Copyright © 2016 John Wiley & Sons, Ltd.
Conflict of interest statement
The authors declared no conflicts of interest
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

