Simulated Respiratory Secretion for Use in the Development of Influenza Diagnostic Assays
- PMID: 27870895
- PMCID: PMC5117718
- DOI: 10.1371/journal.pone.0166800
Simulated Respiratory Secretion for Use in the Development of Influenza Diagnostic Assays
Abstract
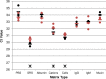
Many assays have been developed for the detection of influenza virus which is an important respiratory pathogen. Development of these assays commonly involves the use of human clinical samples for validation of their performance. However, clinical samples can be difficult to obtain, deteriorate over time, and be inconsistent in composition. The goal of this study was to develop a simulated respiratory secretion (SRS) that could act as a surrogate for clinical samples. To this end, we determined the effects major respiratory secretion components (Na+, K+, Ca2+, cells, albumin IgG, IgM, and mucin) have on the performance of influenza assays including both nucleic acid amplification and rapid antigen assays. Minimal effects on the molecular assays were observed for all of the components tested, except for serum derived human IgG, which suppressed the signal of the rapid antigen assays. Using dot blots we were able to show anti-influenza nucleoprotein IgG antibodies are common in human respiratory samples. We composed a SRS that contained mid-point levels of human respiratory sample components and studied its effect compared to phosphate buffered saline and virus negative clinical sample matrix on the Veritor, Sofia, CDC RT-PCR, Simplexa, cobas Liat, and Alere i influenza assays. Our results demonstrated that a SRS can interact with a variety of test methods in a similar manner to clinical samples with a similar impact on test performance.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
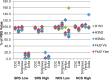

References
-
- Biewenga J, Stoop AE, Baker HE, Swart SJ, Nauta JJ, van Kamp GJ, et al. Nasal secretions from patients with polyps and healthy individuals, collected with a new aspiration system: evaluation of total protein and immunoglobulin concentrations. Ann Clin Biochem 1991. May;28 (Pt 3)(Pt 3):260–266. - PubMed
-
- Johansson SG, Deuschl H. Immunoglobulins in nasal secretion with special reference to IgE. I. Methodological studies. Int Arch Allergy Appl Immunol 1976;52(1–4):364–375. - PubMed
-
- Lorin MI, Gaerlan PF, Mandel ID. Quantitative composition of nasal secretions in normal subjects. J Lab Clin Med 1972. August;80(2):275–281. - PubMed
-
- Page J, Class J, Chapman N, Scholl D. Comparison of flocked swabs versus mattress swabs and nasal aspirates for the collection of nasopharyngeal samples, abstr M-38 In: Specter S, editor. Abstr 23rd Ann Clin Virol Symposium. Clearwater Beach FL ed. Clearwater Beach, FL: American Society for Microbiology; 2007. p. M-38.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

