Dynamic culture substrate that captures a specific extracellular matrix protein in response to light
- PMID: 27877416
- PMCID: PMC5090494
- DOI: 10.1088/1468-6996/12/4/044608
Dynamic culture substrate that captures a specific extracellular matrix protein in response to light
Abstract
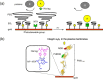
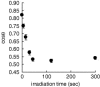
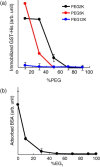
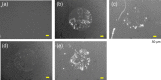
The development of methods for the off-on switching of immobilization or presentation of cell-adhesive peptides and proteins during cell culture is important because such surfaces are useful for the analysis of the dynamic processes of cell adhesion and migration. This paper describes a chemically functionalized gold substrate that captures a genetically tagged extracellular matrix protein in response to light. The substrate was composed of mixed self-assembled monolayers (SAMs) of three disulfide compounds containing (i) a photocleavable poly(ethylene glycol) (PEG), (ii) nitrilotriacetic acid (NTA) and (iii) hepta(ethylene glycol) (EG7). Although the NTA group has an intrinsic high affinity for oligohistidine tag (His-tag) sequences in its Ni2+-ion complex, the interaction was suppressed by the steric hindrance of coexisting PEG on the substrate surface. Upon photoirradiation of the substrate to release the PEG chain from the surface, this interaction became possible and hence the protein was captured at the irradiated regions, while keeping the non-specific adsorption of non-His-tagged proteins blocked by the EG7 underbrush. In this way, we selectively immobilized a His-tagged fibronectin fragment (FNIII7-10) to the irradiated regions. In contrast, when bovine serum albumin-a major serum protein-was added as a non-His-tagged protein, the surface did not permit its capture, with or without irradiation. In agreement with these results, cells were selectively attached to the irradiated patterns only when a His-tagged FNIII7-10 was added to the medium. These results indicate that the present method is useful for studying the cellular behavior on the specific extracellular matrix protein in cell-culturing environments.
Keywords: His-tag; fibronectin; integrin; patterning; photocleavage reaction.
Figures




Similar articles
-
Development and characterization of Ni-NTA-bearing microspheres.Cytometry. 2002 Jul 1;48(3):136-45. doi: 10.1002/cyto.10124. Cytometry. 2002. PMID: 12116359
-
XPS, TOF-SIMS, NEXAFS, and SPR characterization of nitrilotriacetic acid-terminated self-assembled monolayers for controllable immobilization of proteins.Anal Chem. 2008 Apr 1;80(7):2564-73. doi: 10.1021/ac702380w. Epub 2008 Feb 27. Anal Chem. 2008. PMID: 18302347 Free PMC article.
-
NTA-Functionalized Poly(L-lysine)-g-Poly(Ethylene Glycol): A Polymeric Interface for Binding and Studying 6 His-tagged Proteins.Conf Proc IEEE Eng Med Biol Soc. 2005;2006:1036-8. doi: 10.1109/IEMBS.2005.1616595. Conf Proc IEEE Eng Med Biol Soc. 2005. PMID: 17282364
-
Dynamic substrate based on photocleavable poly(ethylene glycol): zeta potential determines the capability of geometrical cell confinement.Langmuir. 2013 Jun 18;29(24):7300-8. doi: 10.1021/la304569e. Epub 2013 Jan 24. Langmuir. 2013. PMID: 23298202
-
Integrins in cell adhesion and signaling.Hum Cell. 1996 Sep;9(3):181-6. Hum Cell. 1996. PMID: 9183647 Review.
Cited by
-
Switching specific biomolecular interactions on surfaces under complex biological conditions.Analyst. 2014 Nov 7;139(21):5400-8. doi: 10.1039/c4an01225a. Analyst. 2014. PMID: 25180245 Free PMC article.
-
Photoactivatable surfaces resolve the impact of gravity vector on collective cell migratory characteristics.Sci Technol Adv Mater. 2023 May 3;24(1):2206525. doi: 10.1080/14686996.2023.2206525. eCollection 2023. Sci Technol Adv Mater. 2023. PMID: 37151805 Free PMC article.
-
Engineering Dynamic Biointerfaces.Curr Opin Chem Eng. 2016 Mar;11:28-33. doi: 10.1016/j.coche.2015.11.005. Curr Opin Chem Eng. 2016. PMID: 26726308 Free PMC article.
-
Electrically-driven modulation of surface-grafted RGD peptides for manipulation of cell adhesion.Chem Commun (Camb). 2014 Dec 21;50(98):15589-92. doi: 10.1039/c4cc06649a. Epub 2014 Oct 31. Chem Commun (Camb). 2014. PMID: 25360452 Free PMC article.
-
4D Self-Morphing Culture Substrate for Modulating Cell Differentiation.Adv Sci (Weinh). 2020 Feb 18;7(6):1902403. doi: 10.1002/advs.201902403. eCollection 2020 Mar. Adv Sci (Weinh). 2020. PMID: 32195081 Free PMC article.
References
-
- Meredith J C, Karim A. and Amis E J. MRS Bull. 2002;27:330. doi: 10.1557/mrs2002.101. - DOI
LinkOut - more resources
Full Text Sources
Research Materials
