Micrometer-level naked-eye detection of caesium particulates in the solid state
- PMID: 27877560
- PMCID: PMC5090578
- DOI: 10.1088/1468-6996/14/1/015002
Micrometer-level naked-eye detection of caesium particulates in the solid state
Abstract
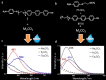
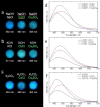
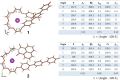
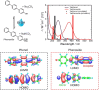
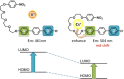
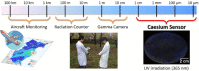
Large amounts of radioactive material were released from the Fukushima Daiichi nuclear plant in Japan, contaminating the local environment. During the early stages of such nuclear accidents, iodine I-131 (half-life 8.02 d) is usually detectable in the surrounding atmosphere and bodies of water. On the other hand, in the long-term, soil and water contamination by Cs-137, which has a half-life of 30.17 years, is a serious problem. In Japan, the government is planning and carrying out radioactive decontamination operations not only with public agencies but also non-governmental organizations, making radiation measurements within Japan. If caesium (also radiocaesium) could be detected by the naked eye then its environmental remediation would be facilitated. Supramolecular material approaches, such as host-guest chemistry, are useful in the design of high-resolution molecular sensors and can be used to convert molecular-recognition processes into optical signals. In this work, we have developed molecular materials (here, phenols) as an optical probe for caesium cation-containing particles with implementation based on simple spray-on reagents and a commonly available fluorescent lamp for naked-eye detection in the solid state. This chemical optical probe provides a higher spatial resolution than existing radioscopes and gamma-ray cameras.
Keywords: 10.11; caesium cation recognition; charge transfer; optical probe; radiocaesium; supramolecular.
Figures
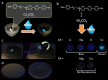
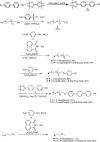
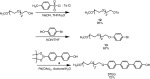
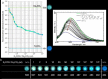


Similar articles
-
Linking heterogeneous distribution of radiocaesium in soils and pond sediments in the Fukushima Daiichi exclusion zone to mobility and potential bioavailability.J Environ Radioact. 2020 Jan;211:106080. doi: 10.1016/j.jenvrad.2019.106080. Epub 2019 Oct 31. J Environ Radioact. 2020. PMID: 31677432
-
Evaluation of the cause of unexplained radiocaesium contamination of brown rice in Fukushima in 2013 using autoradiography and gamma-ray spectrometry.Sci Rep. 2016 Feb 4;6:20386. doi: 10.1038/srep20386. Sci Rep. 2016. PMID: 26843000 Free PMC article.
-
Global and local cancer risks after the Fukushima Nuclear Power Plant accident as seen from Chernobyl: a modeling study for radiocaesium ((134)Cs &(137)Cs).Environ Int. 2014 Mar;64:17-27. doi: 10.1016/j.envint.2013.11.020. Epub 2013 Dec 20. Environ Int. 2014. PMID: 24361922
-
Application of synchrotron radiation and other techniques in analysis of radioactive microparticles emitted from the Fukushima Daiichi Nuclear Power Plant accident-A review.J Environ Radioact. 2019 Jan;196:29-39. doi: 10.1016/j.jenvrad.2018.10.013. Epub 2018 Oct 25. J Environ Radioact. 2019. PMID: 30388426 Review.
-
Radiocesium transfer from hillslopes to the Pacific Ocean after the Fukushima Nuclear Power Plant accident: A review.J Environ Radioact. 2015 Oct;148:92-110. doi: 10.1016/j.jenvrad.2015.06.018. Epub 2015 Jul 3. J Environ Radioact. 2015. PMID: 26142817 Review.
Cited by
-
Celebrating Dr. Katsuhiko Ariga's 60th birthday: from Nanotechnology to Nanoarchitectonics.Sci Technol Adv Mater. 2025 Aug 1;26(1):2532290. doi: 10.1080/14686996.2025.2532290. eCollection 2025. Sci Technol Adv Mater. 2025. PMID: 40761807 Free PMC article. No abstract available.
-
Bio-interactive nanoarchitectonics with two-dimensional materials and environments.Sci Technol Adv Mater. 2022 Mar 30;23(1):199-224. doi: 10.1080/14686996.2022.2054666. eCollection 2022. Sci Technol Adv Mater. 2022. PMID: 35370475 Free PMC article. Review.
-
β-Cyclodextrin/Triclosan Complex-Grafted Methacrylated Glycol Chitosan Hydorgel by Photocrosslinking via Visible Light Irradiation for a Tissue Bio-Adhesive.Int J Mol Sci. 2021 Jan 12;22(2):700. doi: 10.3390/ijms22020700. Int J Mol Sci. 2021. PMID: 33445775 Free PMC article.
-
Improvement of Cs detection performance and formation of CsCl and Cs nanoparticles by tuning graphene oxide quantum dot-based nanocomposite.RSC Adv. 2022 Jul 6;12(30):19667-19677. doi: 10.1039/d2ra02091b. eCollection 2022 Jun 29. RSC Adv. 2022. PMID: 35865579 Free PMC article.
-
Cesium Cation Complexation by a Flavin Receptor via Self-Assembly and Deprotonation.ACS Omega. 2020 Aug 14;5(33):21226-21230. doi: 10.1021/acsomega.0c03006. eCollection 2020 Aug 25. ACS Omega. 2020. PMID: 32875259 Free PMC article.
References
-
- Essen L. and Parry J V L. Nature. 1995;176:280. doi: 10.1038/176280a0. - DOI
-
- Butterman W C, Brooks W E, Reese R G., Jr . Open-File Report. Virginia: US Geological Survey; 2004. Mineral commodity profile: caesium; p. p 5. 2004–1432.
-
- International Atomic Energy Agency 2006 Environmental consequences of the Chernobyl accident and their remediation: twenty years of experience Report of the UN Chernobyl Forum Expert Group ‘Environment’ (Vienna: IAEA) STI/PUB/1239
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
