Activation of Both the Calpain and Ubiquitin-Proteasome Systems Contributes to Septic Cardiomyopathy through Dystrophin Loss/Disruption and mTOR Inhibition
- PMID: 27880847
- PMCID: PMC5120800
- DOI: 10.1371/journal.pone.0166839
Activation of Both the Calpain and Ubiquitin-Proteasome Systems Contributes to Septic Cardiomyopathy through Dystrophin Loss/Disruption and mTOR Inhibition
Abstract
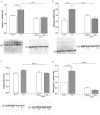
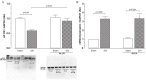
Cardiac dysfunction caused by the impairment of myocardial contractility has been recognized as an important factor contributing to the high mortality in sepsis. Calpain activation in the heart takes place in response to increased intracellular calcium influx resulting in proteolysis of structural and contractile proteins with subsequent myocardial dysfunction. The purpose of the present study was to test the hypothesis that increased levels of calpain in the septic heart leads to disruption of structural and contractile proteins and that administration of calpain inhibitor-1 (N-acetyl-leucinyl-leucinyl-norleucinal (ALLN)) after sepsis induced by cecal ligation and puncture prevents cardiac protein degradation. We also tested the hypothesis that calpain plays a role in the modulation of protein synthesis/degradation through the activation of proteasome-dependent proteolysis and inhibition of the mTOR pathway. Severe sepsis significantly increased heart calpain-1 levels and promoted ubiquitin and Pa28β over-expression with a reduction in the mTOR levels. In addition, sepsis reduced the expression of structural proteins dystrophin and β-dystroglycan as well as the contractile proteins actin and myosin. ALLN administration prevented sepsis-induced increases in calpain and ubiquitin levels in the heart, which resulted in decreased of structural and contractile proteins degradation and basal mTOR expression levels were re-established. Our results support the concept that increased calpain concentrations may be part of an important mechanism of sepsis-induced cardiac muscle proteolysis.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures



Similar articles
-
Disruption of calcium homeostasis in cardiomyocytes underlies cardiac structural and functional changes in severe sepsis.PLoS One. 2013 Jul 23;8(7):e68809. doi: 10.1371/journal.pone.0068809. Print 2013. PLoS One. 2013. PMID: 23935889 Free PMC article.
-
Calpain-mediated dystrophin disruption may be a potential structural culprit behind chronic doxorubicin-induced cardiomyopathy.Eur J Pharmacol. 2011 Nov 30;670(2-3):541-53. doi: 10.1016/j.ejphar.2011.09.021. Epub 2011 Sep 21. Eur J Pharmacol. 2011. PMID: 21946105
-
Cardiomyocyte degeneration with calpain deficiency reveals a critical role in protein homeostasis.Circ Res. 2007 Apr 13;100(7):1071-8. doi: 10.1161/01.RES.0000261938.28365.11. Epub 2007 Mar 1. Circ Res. 2007. PMID: 17332428
-
Cooperation between proteolytic systems in cardiomyocyte recycling.Cardiovasc Res. 2012 Oct 1;96(1):46-52. doi: 10.1093/cvr/cvs236. Epub 2012 Jul 25. Cardiovasc Res. 2012. PMID: 22843702 Review.
-
Build it up-Tear it down: protein quality control in the cardiac sarcomere.Cardiovasc Res. 2009 Feb 15;81(3):439-48. doi: 10.1093/cvr/cvn289. Epub 2008 Oct 29. Cardiovasc Res. 2009. PMID: 18974044 Free PMC article. Review.
Cited by
-
Modulation of the mTOR Pathway by Curcumin in the Heart of Septic Mice.Pharmaceutics. 2022 Oct 24;14(11):2277. doi: 10.3390/pharmaceutics14112277. Pharmaceutics. 2022. PMID: 36365096 Free PMC article.
-
Calcium handling dysfunction and cardiac damage following acute ventricular preload challenge in the dystrophin-deficient mouse heart.Am J Physiol Heart Circ Physiol. 2023 Nov 1;325(5):H1168-H1177. doi: 10.1152/ajpheart.00265.2023. Epub 2023 Sep 22. Am J Physiol Heart Circ Physiol. 2023. PMID: 37737731 Free PMC article.
-
Breakdown of Filamentous Myofibrils by the UPS-Step by Step.Biomolecules. 2021 Jan 15;11(1):110. doi: 10.3390/biom11010110. Biomolecules. 2021. PMID: 33467597 Free PMC article. Review.
-
Cardiac Pathophysiology and the Future of Cardiac Therapies in Duchenne Muscular Dystrophy.Int J Mol Sci. 2019 Aug 22;20(17):4098. doi: 10.3390/ijms20174098. Int J Mol Sci. 2019. PMID: 31443395 Free PMC article. Review.
-
Temporally Distinct Regulation of Pathways Contributing to Cardiac Proteostasis During the Acute and Recovery Phases of Sepsis.Shock. 2018 Dec;50(6):616-626. doi: 10.1097/SHK.0000000000001084. Shock. 2018. PMID: 29240643 Free PMC article.
References
-
- Werdan K, Oelke A, Mueller-Werdan U. Myocardial Depression or Septic Cardiomyopathy in Vincent JL Yearbook of intensive care and emergency medicine. Springer, Heidelberg: 2009;pp 183–194.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

