Disruption of Autophagic Degradation with ROC-325 Antagonizes Renal Cell Carcinoma Pathogenesis
- PMID: 27881580
- PMCID: PMC5593077
- DOI: 10.1158/1078-0432.CCR-16-1742
Disruption of Autophagic Degradation with ROC-325 Antagonizes Renal Cell Carcinoma Pathogenesis
Abstract
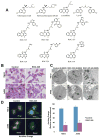
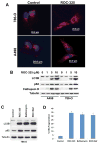
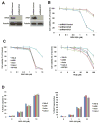
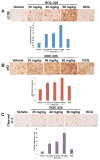
Purpose: Although autophagy plays important roles in malignant pathogenesis and drug resistance, there are few clinical agents that disrupt this pathway, and the potential therapeutic benefit of autophagy inhibition remains undetermined. We used medicinal chemistry approaches to generate a series of novel agents that inhibit autophagic degradation.Experimental Design: ROC-325 was selected as a lead compound for further evaluation. Comprehensive in vitro and in vivo studies were conducted to evaluate the selectivity, tolerability, and efficacy of ROC-325 in preclinical models of renal cell carcinoma (RCC) with HCQ serving as a comparator. Markers of autophagy inhibition and cell death were evaluated in tumor specimens.Results: ROC-325 exhibited superior in vitro anticancer effects compared with the existing autophagy inhibitor hydroxychloroquine (HCQ) in 12 different cancer cell lines with diverse genetic backgrounds. Focused studies of the mechanism of action and efficacy of ROC-325 in RCC cells showed that drug treatment induced hallmark characteristics of autophagy inhibition, including accumulation of autophagosomes with undegraded cargo, lysosomal deacidification, p62 stabilization, and disruption of autophagic flux. Subsequent experiments showed that ROC-325 antagonized RCC growth and survival in an ATG5/7-dependent manner, induced apoptosis, and exhibited favorable selectivity. Oral administration of ROC-325 to mice bearing 786-0 RCC xenografts was well tolerated, was significantly more effective at inhibiting tumor progression than HCQ, and inhibited autophagy in vivoConclusions: Our findings demonstrate that ROC-325 has superior preclinical anticancer activity compared with HCQ and support the clinical investigation of its safety and preliminary efficacy in patients with RCC and other autophagy-dependent malignancies. Clin Cancer Res; 23(11); 2869-79. ©2016 AACR.
©2016 American Association for Cancer Research.
Conflict of interest statement
Figures


References
-
- Janku F, McConkey DJ, Hong DS, Kurzrock R. Autophagy as a target for anticancer therapy. Nat Rev Clin Oncol. 2011;8:528–39. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

