Nucleoside-Diphosphate-Kinase of P. gingivalis is Secreted from Epithelial Cells In the Absence of a Leader Sequence Through a Pannexin-1 Interactome
- PMID: 27883084
- PMCID: PMC5121656
- DOI: 10.1038/srep37643
Nucleoside-Diphosphate-Kinase of P. gingivalis is Secreted from Epithelial Cells In the Absence of a Leader Sequence Through a Pannexin-1 Interactome
Abstract
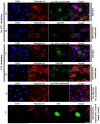
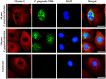
Nucleoside-diphosphate-kinases (NDKs) are leaderless, multifunctional enzymes. The mode(s) of NDK secretion is currently undefined, while extracellular translocation of bacterial NDKs is critical for avoidance of host pathogen clearance by opportunistic pathogens such as Porphyromonas gingivalis. P. gingivalis-NDK during infection inhibits extracellular-ATP (eATP)/P2X7-receptor mediated cell death in gingival epithelial cells (GECs) via eATP hydrolysis. Furthermore, depletion of pannexin-1-hemichannel (PNX1) coupled with P2X7-receptor blocks the infection-induced eATP release in GECs, and P. gingivalis-NDK impacts this pathway. Ultrastructural and confocal microscopy of P. gingivalis-co-cultured GECs or green-fluorescent-protein (GFP)-P. gingivalis-NDK transfected GECs revealed a perinuclear/cytoplasmic localization of NDK. eATP stimulation induced NDK recruitment to the cell periphery. Depletion of PNX1 by siRNA or inhibition by probenecid resulted in significant blocking of extracellular NDK activity and secretion using ATPase and ELISA assays. Co-immunoprecipitation-coupled Mass-spectrometry method revealed association of P. gingivalis-NDK to the myosin-9 motor molecule. Interestingly, inhibition of myosin-9, actin, and lipid-rafts, shown to be involved in PNX1-hemichannel function, resulted in marked intracellular accumulation of NDK and decreased NDK secretion from infected GECs. These results elucidate for the first time PNX1-hemichannels as potentially main extracellular translocation pathway for NDKs from an intracellular pathogen, suggesting that PNX1-hemichannels may represent a therapeutic target for chronic opportunistic infections.
Figures





References
-
- Chakrabarty A. M. Nucleoside diphosphate kinase: role in bacterial growth, virulence, cell signalling and polysaccharide synthesis. Mol Microbiol 28, 875–882 (1998). - PubMed
-
- Lascu L., Giartosio A., Ransac S. & Erent M. Quaternary structure of nucleoside diphosphate kinases. J Bioenerg Biomembr 32, 227–236 (2000). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

