Anti-LRP/LR-specific antibody IgG1-iS18 impedes adhesion and invasion of pancreatic cancer and neuroblastoma cells
- PMID: 27884119
- PMCID: PMC5123428
- DOI: 10.1186/s12885-016-2953-2
Anti-LRP/LR-specific antibody IgG1-iS18 impedes adhesion and invasion of pancreatic cancer and neuroblastoma cells
Abstract
Background: Cancer has become a global burden due to its high incidence and mortality rates, with an estimated 14.1 million cancer cases reported worldwide in 2012 particularly as a result of metastasis. Metastasis involves two crucial steps: adhesion and invasion, and the non-integrin receptor; the 37-kDa/67-kDa laminin receptor precursor/ high affinity laminin receptor (LRP/LR) has been shown to be overexpressed on the surface of tumorigenic cells, thus being implicated in the enhancement of these two crucial steps. The current study investigated the role of LRP/LR on the aggressiveness of pancreatic cancer (AsPC-1) and neuroblastoma (IMR-32) cells with respect to their adhesive and invasive potential.
Methods: AsPC-1 and IMR-32 cells were utilized as the experimental cell lines for the study. Cell surface LRP/LR levels were visualised and quantified on the experimental and control (MCF-7) cell lines via confocal microscopy and flow cytometry, respectively. Total LRP/LR levels in the cell lines were assessed by Western blotting and the adhesive and invasive potential of the above-mentioned cell lines was determined before and after supplementation with the anti-LRP/LR specific antibody IgG1-iS18. Statistical significance of the data was confirmed via the use of the two-tailed student's t-test and Pearson's correlation coefficient.
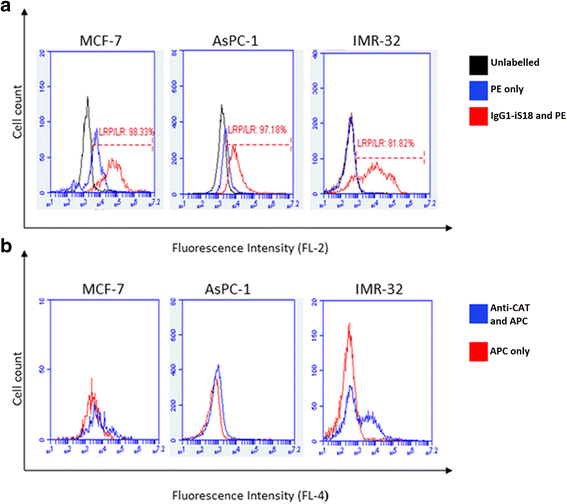
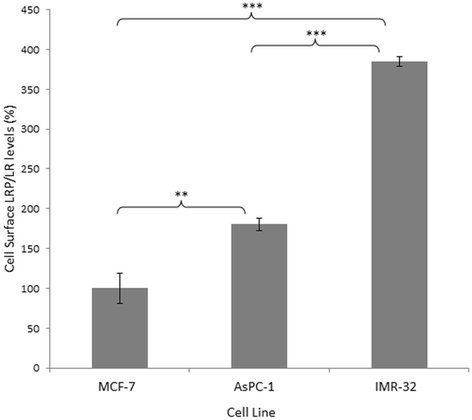
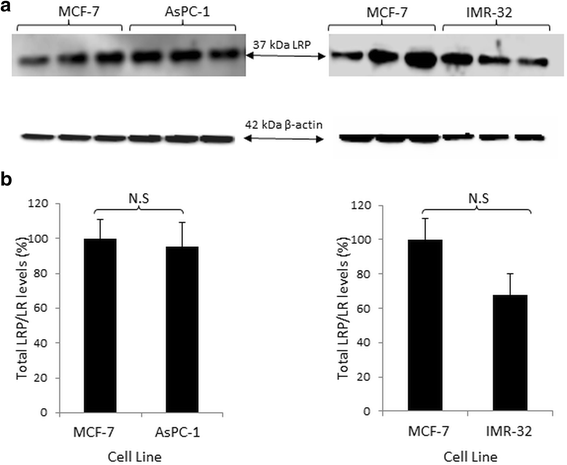
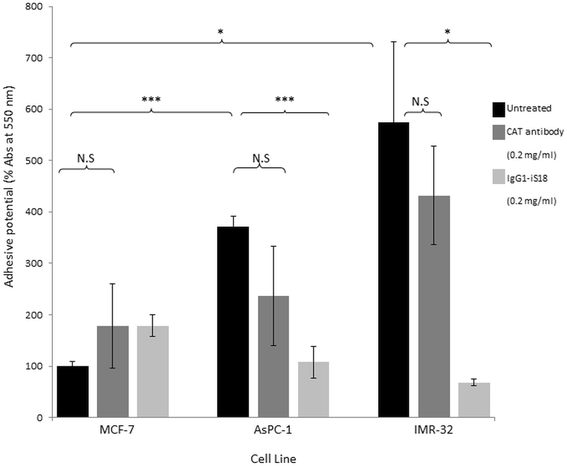
Results: Flow cytometry revealed that AsPC-1 and IMR-32 cells displayed significantly higher cell surface LRP/LR levels in comparison to the MCF-7 control cell line. However, Western blotting and subsequent densitometric analysis revealed that all three tumorigenic cell lines displayed no significant difference in total LRP/LR levels. The treatment of AsPC-1 and IMR-32 cells with IgG1-iS18 caused a significant reduction in the adhesive and invasive potential of the cells to laminin-1 and through the ECM-like Matrigel™, respectively. Pearson's correlation coefficients indicated a high correlation, thus suggesting a directly proportional relationship between cell surface LRP/LR levels and the adhesive and invasive potential of AsPC-1 and IMR-32 cells.
Conclusion: These findings suggest that through the interference of the LRP/LR-laminin-1 interaction, the anti-LRP/LR specific antibody IgG1-iS18 may act as an alternative therapeutic tool for the treatment of metastatic pancreatic cancer and neuroblastoma.
Keywords: 37 kDa/67-kDa laminin receptor (LRP/LR); Adhesion; Invasion; Laminin-1; Metastasis; Neuroblastoma; Pancreatic cancer.
Figures


Similar articles
-
IgG1-iS18 impedes the adhesive and invasive potential of early and late stage malignant melanoma cells.Exp Cell Res. 2017 Feb 15;351(2):135-141. doi: 10.1016/j.yexcr.2017.01.009. Epub 2017 Jan 22. Exp Cell Res. 2017. PMID: 28118986
-
Anti-LRP/LR specific antibody IgG1-iS18 impedes adhesion and invasion of liver cancer cells.PLoS One. 2014 May 5;9(5):e96268. doi: 10.1371/journal.pone.0096268. eCollection 2014. PLoS One. 2014. PMID: 24798101 Free PMC article.
-
Anti-LRP/LR-specific antibody IgG1-iS18 significantly reduces adhesion and invasion of metastatic lung, cervix, colon and prostate cancer cells.J Mol Biol. 2012 May 25;419(1-2):102-9. doi: 10.1016/j.jmb.2012.02.035. Epub 2012 Mar 3. J Mol Biol. 2012. PMID: 22391421
-
Patented therapeutic approaches targeting LRP/LR for cancer treatment.Expert Opin Ther Pat. 2019 Dec;29(12):987-1009. doi: 10.1080/13543776.2019.1693543. Epub 2019 Nov 20. Expert Opin Ther Pat. 2019. PMID: 31722579 Review.
-
Patented biological approaches for the therapeutic modulation of the 37 kDa/67 kDa laminin receptor.Expert Opin Ther Pat. 2011 Jan;21(1):35-53. doi: 10.1517/13543776.2011.539203. Epub 2010 Nov 27. Expert Opin Ther Pat. 2011. PMID: 21110766 Review.
Cited by
-
Targeting the Extra-Cellular Matrix-Tumor Cell Crosstalk for Anti-Cancer Therapy: Emerging Alternatives to Integrin Inhibitors.Front Oncol. 2020 Jul 22;10:1231. doi: 10.3389/fonc.2020.01231. eCollection 2020. Front Oncol. 2020. PMID: 32793493 Free PMC article. Review.
-
Non-integrin laminin receptor (LamR) plays a role in axonal outgrowth from chicken DRG via modulating the Akt and Erk signaling.Front Cell Dev Biol. 2024 Jul 31;12:1433947. doi: 10.3389/fcell.2024.1433947. eCollection 2024. Front Cell Dev Biol. 2024. PMID: 39144252 Free PMC article.
-
Knock-down of LRP/LR influences signalling pathways in late-stage colorectal carcinoma cells.BMC Cancer. 2021 Apr 9;21(1):392. doi: 10.1186/s12885-021-08081-3. BMC Cancer. 2021. PMID: 33836696 Free PMC article.
-
Channeling the Natural Properties of Sindbis Alphavirus for Targeted Tumor Therapy.Int J Mol Sci. 2023 Oct 6;24(19):14948. doi: 10.3390/ijms241914948. Int J Mol Sci. 2023. PMID: 37834397 Free PMC article. Review.
-
37 kDa LRP::FLAG enhances telomerase activity and reduces senescent markers in vitro.Oncotarget. 2017 Sep 27;8(49):86646-86656. doi: 10.18632/oncotarget.21278. eCollection 2017 Oct 17. Oncotarget. 2017. PMID: 29156824 Free PMC article.
References
-
- Worldwide data. http://www.wcrf.org/int/cancer-facts-figures/worldwide-data. Accessed 7 Apr 2015.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

